4,5,7,8,17-Penta-hydr-oxy-14,18-dimethyl-6-methyl-ene-3,10-dioxapenta-cyclo-[9.8.0.0.0.0]nona-dec-14-ene-9,16-dione methanol solvate dihydrate
- PMID: 21582604
- PMCID: PMC2968991
- DOI: 10.1107/S1600536809010502
4,5,7,8,17-Penta-hydr-oxy-14,18-dimethyl-6-methyl-ene-3,10-dioxapenta-cyclo-[9.8.0.0.0.0]nona-dec-14-ene-9,16-dione methanol solvate dihydrate
Abstract
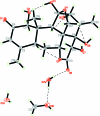
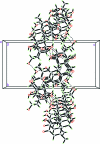
The title quassinoid compound, C(20)H(24)O(9)·CH(3)OH·2H(2)O, is a natural eurycomanone isolated from the roots of Eurycoma longifolia. The mol-ecules contain a fused five-ring system, with one tetra-hydro-furan ring adopting an envelope conformation, one tetra-hydro-pyran-2-one ring in a screw boat conformation, one cyclo-hexenone ring in a half-chair conformation and two cyclo-hexane rings in chair conformations. Intra-molecular C-H⋯O inter-actions generate S(5) ring motifs and an O-H⋯O inter-action generates an S(7) ring motif. In the crystal, mol-ecules are linked via inter-molecular O-H⋯O inter-actions along the b axis and further stacked along a axis. The absolute configuration of the title compound was inferred from previously solved structures of its analogues.
Figures
References
-
- Allen, F. H., Kennard, O., Watson, D. G., Brammer, L., Orpen, A. G. & Taylor, R. (1987). J. Chem. Soc. Perkin Trans. 2, pp. S1–19.
-
- Ang, H. H., Chan, K. L. & Mak, J. W. (1995). Planta Med 61, 177–178. - PubMed
-
- Bernstein, J., Davis, R. E., Shimoni, L. & Chang, N.-L. (1995). Angew. Chem. Int. Ed. Engl.34, 1555–1573.
-
- Bruker (2005). APEX2, SAINT and SADABS Bruker AXS Inc., Madison, Wisconsin, USA.
-
- Chan, K.-L., Choo, C.-Y., Abdullah, N. R. & Ismail, Z. (2004). J. Ethnopharmacol 92, 223–227. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
