High molecular weight kininogen activates B2 receptor signaling pathway in human vascular endothelial cells
- PMID: 21586566
- PMCID: PMC3137031
- DOI: 10.1074/jbc.M110.211557
High molecular weight kininogen activates B2 receptor signaling pathway in human vascular endothelial cells
Abstract
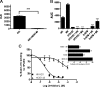
The nonenzymatic cofactor high molecular weight kininogen (HK) is a precursor of bradykinin (BK). The production of BK from HK by plasma kallikrein has been implicated in the pathogenesis of inflammation and vascular injury. However, the functional role of HK in the absence of prekallikrein (PK), the proenzyme of plasma kallikrein, on vascular endothelial cells is not fully defined. In addition, no clinical abnormality is seen in PK-deficient patients. Therefore, an investigation into the effect of HK, in the absence of PK, on human pulmonary artery endothelial cell (HPAEC) function was performed. HK caused a marked and dose-dependent increase in the intracellular calcium [Ca(2+)](i) level in HPAEC. Gd(3+) and verapamil potentiated the HK-induced increase in [Ca(2+)](i). HK-induced Ca(2+) increase stimulated endothelial nitric oxide (NO) and prostacyclin (PGI(2)) production. The inhibitors of B(2) receptor-dependent signaling pathway impaired HK-mediated signal transduction in HPAEC. HK had no effect on endothelial permeability at physiological concentration. This study demonstrated that HK regulates endothelial cell function. HK could play an important role in maintaining normal endothelial function and blood flow and serve as a cardioprotective peptide.
Figures

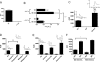
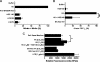

References
-
- Ma J. X., Song Q., Hatcher H. C., Crouch R. K., Chao L., Chao J. (1996) Exp. Eye Res. 63, 19–26 - PubMed
-
- Takano M., Kondo J., Yayama K., Otani M., Sano K., Okamoto H. (1997) Biochim. Biophys. Acta. 1352, 222–230 - PubMed
-
- Bior A. D., Pixley R. A., Colman R. W. (2007) J. Thromb. Haemost. 5, 403–411 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

