(Z)-3-(2-{2-[1-(4-Hy-droxy-phen-yl)ethyl-idene]hydrazin-1-yl}-1,3-thia-zol-4-yl)-2H-chromen-2-one
- PMID: 21587863
- PMCID: PMC3006946
- DOI: 10.1107/S1600536810021604
(Z)-3-(2-{2-[1-(4-Hy-droxy-phen-yl)ethyl-idene]hydrazin-1-yl}-1,3-thia-zol-4-yl)-2H-chromen-2-one
Abstract
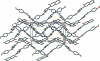
In the title compound, C(20)H(15)N(3)O(3)S, an intra-molecular C-H⋯O hydrogen bond generates an S(6) ring motif. The chromene ring system is inclined at dihedral angles of 14.21 (9) and 9.91 (10)°, respectively, with respect to the thia-zole and benzene rings. The thia-zole ring makes a dihedral angle of 24.06 (11)° with the benzene ring. In the crystal structure, O-H⋯O hydrogen bonds link the mol-ecules into a zigzag chain along [20]. Weak N-H⋯O and C-H⋯O inter-actions connect the chains into a three-dimensional network. π-π stacking inter-actions with a centroid-centroid distance of 3.4209 (14) Å are also observed between the chains.
Figures
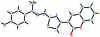
References
-
- Allen, F. H., Kennard, O., Watson, D. G., Brammer, L., Orpen, A. G. & Taylor, R. (1987). J. Chem. Soc. Perkin Trans. 2, pp. S1–19.
-
- Anderson, D. M., Shelley, S., Crick, N. & Buraglio, L. (2002). J. Clin. Pharmacol.42, 1358–1365. - PubMed
-
- Bernstein, J., Davis, R. E., Shimoni, L. & Chang, N.-L. (1995). Angew. Chem. Int. Ed. Engl.34, 1555–1573.
-
- Bruker (2009). APEX2, SAINT and SADABS Bruker AXS Inc., Madison, Wisconsin, USA.
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
