mTORC2 protein complex-mediated Akt (Protein Kinase B) Serine 473 Phosphorylation is not required for Akt1 activity in human platelets [corrected]
- PMID: 21592956
- PMCID: PMC3137030
- DOI: 10.1074/jbc.M110.202341
mTORC2 protein complex-mediated Akt (Protein Kinase B) Serine 473 Phosphorylation is not required for Akt1 activity in human platelets [corrected]
Erratum in
- J Biol Chem. 2011 Sep 2;286(35):31062
Abstract
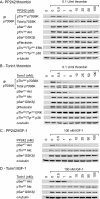
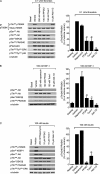
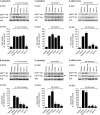
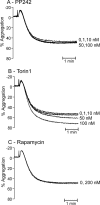
Protein kinase B (PKB, Akt) is a Ser/Thr kinase involved in the regulation of cell survival, proliferation, and metabolism and is activated by dual phosphorylation on Thr(308) in the activation loop and Ser(473) in the hydrophobic motif. It plays a contributory role to platelet function, although little is known about its regulation. In this study, we investigated the role of the mammalian target of rapamycin complex (mTORC)-2 in Akt regulation using the recently identified small molecule ATP competitive mTOR inhibitors PP242 and Torin1. Both PP242 and Torin1 blocked thrombin and insulin-like growth factor 1-mediated Akt Ser(473) phosphorylation with an IC(50) between 1 and 5 nm, whereas the mTORC1 inhibitor rapamycin had no effect. Interestingly, PP242 and Torin1 had no effect on Akt Thr(308) phosphorylation, Akt1 activity, and phosphorylation of the Akt substrate glycogen synthase kinase 3β, indicating that Ser(473) phosphorylation is not necessary for Thr(308) phosphorylation and maximal Akt1 activity. In contrast, Akt2 activity was significantly reduced, concurrent with inhibition of PRAS40 phosphorylation, in the presence of PP242 and Torin1. Other signaling pathways, including phospholipase C/PKC and the MAPK pathway, were unaffected by PP242 and Torin1. Together, these results demonstrate that mTORC2 is the kinase that phosphorylates Akt Ser(473) in human platelets but that this phosphorylation is dispensable for Thr(308) phosphorylation and Akt1 activity.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
- PG/08/056/25325/BHF_/British Heart Foundation/United Kingdom
- PG/10/100/28658/BHF_/British Heart Foundation/United Kingdom
- FS/12/3/29232/BHF_/British Heart Foundation/United Kingdom
- FS/05/047/18875/BHF_/British Heart Foundation/United Kingdom
- PG/08/113/25515/BHF_/British Heart Foundation/United Kingdom
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

