Growth of engineered human myocardium with mechanical loading and vascular coculture
- PMID: 21597009
- PMCID: PMC3140796
- DOI: 10.1161/CIRCRESAHA.110.237206
Growth of engineered human myocardium with mechanical loading and vascular coculture
Abstract
Rationale: The developing heart requires both mechanical load and vascularization to reach its proper size, yet the regulation of human heart growth by these processes is poorly understood.
Objective: We seek to elucidate the responses of immature human myocardium to mechanical load and vascularization using tissue engineering approaches.
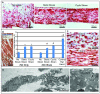
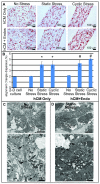
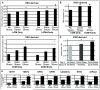
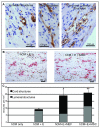
Methods and results: Using human embryonic stem cell and human induced pluripotent stem cell-derived cardiomyocytes in a 3-dimensional collagen matrix, we show that uniaxial mechanical stress conditioning promotes 2-fold increases in cardiomyocyte and matrix fiber alignment and enhances myofibrillogenesis and sarcomeric banding. Furthermore, cyclic stress conditioning markedly increases cardiomyocyte hypertrophy (2.2-fold) and proliferation rates (21%) versus unconditioned constructs. Addition of endothelial cells enhances cardiomyocyte proliferation under all stress conditions (14% to 19%), and addition of stromal supporting cells enhances formation of vessel-like structures by ≈10-fold. Furthermore, these optimized human cardiac tissue constructs generate Starling curves, increasing their active force in response to increased resting length. When transplanted onto hearts of athymic rats, the human myocardium survives and forms grafts closely apposed to host myocardium. The grafts contain human microvessels that are perfused by the host coronary circulation.
Conclusions: Our results indicate that both mechanical load and vascular cell coculture control cardiomyocyte proliferation, and that mechanical load further controls the hypertrophy and architecture of engineered human myocardium. Such constructs may be useful for studying human cardiac development as well as for regenerative therapy.
Figures



Comment in
-
The beat goes on: human heart muscle from pluripotent stem cells.Circ Res. 2011 Jun 24;109(1):2-4. doi: 10.1161/CIRCRESAHA.111.248039. Circ Res. 2011. PMID: 21700948 No abstract available.
References
-
- Montgomery MO, Jiao Y, Phillips SJ, Singh G, Xu J, Balsara R, Litvin J. Alterations in sheep fetal right ventricular tissue with induced hemodynamic pressure overload. Basic Res Cardiol. 1998;93:192–200. - PubMed
-
- Tobita K, Keller BB. Right and left ventricular wall deformation patterns in normal and left heart hypoplasia chick embryos. Am J Physiol Heart Circ Physiol. 2000;279:H959–969. - PubMed
-
- Voronov DA, Alford PW, Xu G, Taber LA. The role of mechanical forces in dextral rotation during cardiac looping in the chick embryo. Dev Biol. 2004;272:339–350. - PubMed
-
- Kira Y, Nakaoka T, Hashimoto E, Okabe F, Asano S, Sekine I. Effect of long-term cyclic mechanical load on protein synthesis and morphological changes in cultured myocardial cells from neonatal rat. Cardiovasc Drugs Ther. 1994;8:251–262. - PubMed
-
- Kuruvilla L, Kartha CC. Molecular mechanisms in endothelial regulation of cardiac function. Mol Cell Biochem. 2003;253:113–123. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

