Pathological heterogeneity in amyotrophic lateral sclerosis with FUS mutations: two distinct patterns correlating with disease severity and mutation
- PMID: 21604077
- PMCID: PMC3319073
- DOI: 10.1007/s00401-011-0838-7
Pathological heterogeneity in amyotrophic lateral sclerosis with FUS mutations: two distinct patterns correlating with disease severity and mutation
Abstract
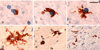
Mutations in the gene encoding the fused in sarcoma (FUS) protein are responsible for ~3% of familial amyotrophic lateral sclerosis (ALS) and <1% of sporadic ALS (ALS-FUS). Descriptions of the associated neuropathology are few and largely restricted to individual case reports. To better define the neuropathology associated with FUS mutations, we have undertaken a detailed comparative analysis of six cases of ALS-FUS that include sporadic and familial cases, with both juvenile and adult onset, and with four different FUS mutations. We found significant pathological heterogeneity among our cases, with two distinct patterns that correlated with the disease severity and the specific mutation. Frequent basophilic inclusions and round FUS-immunoreactive (FUS-ir) neuronal cytoplasmic inclusions (NCI) were a consistent feature of our early-onset cases, including two with the p.P525L mutation. In contrast, our late-onset cases that included two with the p.R521C mutation had tangle-like NCI and numerous FUS-ir glial cytoplasmic inclusions. Double-labeling experiments demonstrated that many of the glial inclusions were in oligodendrocytes. Comparison with the neuropathology of cases of frontotemporal lobar degeneration with FUS-ir pathology showed significant differences and suggests that FUS mutations are associated with a distinct pathobiology.
Figures




References
-
- Aizawa H, Kimura T, Hashimoto K, et al. Basophilic cytoplasmic inclusions in a case of sporadic juvenile amyotrophic lateral sclerosis. J Neurol Sci. 2000;176:109–113. - PubMed
-
- Blair IP, Williams KL, Warrich ST, et al. FUS mutations in amyotrophic lateral sclerosis: clinical, pathological, neurophysiological and genetic analysis. J Neurol Neurosurg Psychiatry. 2010;81:639–645. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

