The hybrid histidine kinase Hk1 is part of a two-component system that is essential for survival of Borrelia burgdorferi in feeding Ixodes scapularis ticks
- PMID: 21606185
- PMCID: PMC3147546
- DOI: 10.1128/IAI.05136-11
The hybrid histidine kinase Hk1 is part of a two-component system that is essential for survival of Borrelia burgdorferi in feeding Ixodes scapularis ticks
Abstract
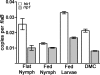
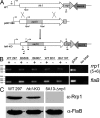
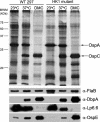
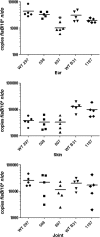
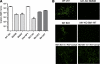
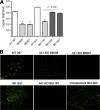
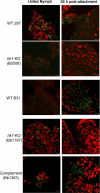
Two-component systems (TCS) are principal mechanisms by which bacteria adapt to their surroundings. Borrelia burgdorferi encodes only two TCS. One is comprised of a histidine kinase, Hk2, and the response regulator Rrp2. While the contribution of Hk2 remains unclear, Rrp2 is part of a regulatory pathway involving the spirochete's alternate sigma factors, RpoN and RpoS. Genes within the Rrp2/RpoN/RpoS regulon function to promote tick transmission and early infection. The other TCS consists of a hybrid histidine kinase, Hk1, and the response regulator Rrp1. Hk1 is composed of two periplasmic sensor domains (D1 and D2), followed by conserved cytoplasmic histidine kinase core, REC, and Hpt domains. In addition to its REC domain, Rrp1 contains a GGDEF motif characteristic of diguanylate cyclases. To investigate the role of Hk1 during the enzootic cycle, we inactivated this gene in two virulent backgrounds. Extensive characterization of the resulting mutants revealed a dramatic phenotype whereby Hk1-deficient spirochetes are virulent in mice and able to migrate out of the bite site during feeding but are killed within the midgut following acquisition. We hypothesize that the phosphorelay between Hk1 and Rrp1 is initiated by the binding of feeding-specific ligand(s) to Hk1 sensor domain D1 and/or D2. Once activated, Rrp1 directs the synthesis of cyclic dimeric GMP (c-di-GMP), which, in turn, modulates the expression and/or activity of gene products required for survival within feeding ticks. In contrast to the Rrp2/RpoN/RpoS pathway, which is active only within feeding nymphs, the Hk1/Rrp1 TCS is essential for survival during both larval and nymphal blood meals.
Figures

References
-
- Arnold K., Bordoli L., Kopp J., Schwede T. 2006. The SWISS-MODEL workspace: a web-based environment for protein structure homology modelling. Bioinformatics 22:195–201 - PubMed
-
- Balashov Y. S. 1972. Blood-sucking ticks (Ixodidae)-vectors of disease of man and animals. Misc. Pub. Entomol. Soc. Am. 8:161–376
-
- Bergstrom S., Zuckert W. R. 2010. Structure, function and biogenesis of the Borrelia cell envelope, p. 139–166 In Samuels D. S., Radolf J. D. (ed.), Borrelia: molecular biology, host interaction and pathogenesis. Calister Academic Press, Norfolk, United Kingdom
-
- Berntsson R. P. A., Smits S. H. J., Schmitt L., Slotboom D.-J., Poolman B. 2010. A structural classification of substrate-binding proteins. FEBS Lett. 584:2606–2617 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R56 AI059373/AI/NIAID NIH HHS/United States
- U54 AI057159/AI/NIAID NIH HHS/United States
- R03 AI085248/AI/NIAID NIH HHS/United States
- R21 AI085310/AI/NIAID NIH HHS/United States
- R01 AI059373/AI/NIAID NIH HHS/United States
- 3R01AI029735-20S1/AI/NIAID NIH HHS/United States
- U54 AI-057159/AI/NIAID NIH HHS/United States
- A1080615/PHS HHS/United States
- AI-29735/AI/NIAID NIH HHS/United States
- R56 AI029735/AI/NIAID NIH HHS/United States
- R01 AI029735/AI/NIAID NIH HHS/United States
- AI085310/AI/NIAID NIH HHS/United States
- AI059373/AI/NIAID NIH HHS/United States
- AI085248/AI/NIAID NIH HHS/United States
LinkOut - more resources
Full Text Sources
Miscellaneous

