Urokinase plasminogen activator regulates pulmonary arterial contractility and vascular permeability in mice
- PMID: 21617202
- PMCID: PMC3262683
- DOI: 10.1165/rcmb.2010-0302OC
Urokinase plasminogen activator regulates pulmonary arterial contractility and vascular permeability in mice
Abstract
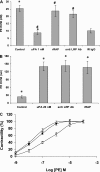
The concentration of urokinase plasminogen activator (uPA) is elevated in pathological settings such as acute lung injury, where pulmonary arterial contractility and permeability are disrupted. uPA limits the accretion of fibrin after injury. Here we investigated whether uPA also regulates pulmonary arterial contractility and permeability. Contractility was measured using isolated pulmonary arterial rings. Pulmonary blood flow was measured in vivo by Doppler and pulmonary vascular permeability, according to the extravasation of Evans blue. Our data show that uPA regulates the in vitro pulmonary arterial contractility induced by phenylephrine in a dose-dependent manner through two receptor-dependent pathways, and regulates vascular contractility and permeability in vivo. Physiological concentrations of uPA (≤1 nM) stimulate the contractility of pulmonary arterial rings induced by phenylephrine through the low-density lipoprotein receptor-related protein receptor. The procontractile effect of uPA is independent of its catalytic activity. At pathophysiological concentrations, uPA (20 nM) inhibits contractility and increases vascular permeability. The inhibition of vascular contractility and increase of vascular permeability is mediated through a two-step process that involves docking to N-methyl-d-aspartate receptor-1 (NMDA-R1) on pulmonary vascular smooth muscle cells, and requires catalytic activity. Peptides that specifically inhibit the docking of uPA to NMDA-R, or the uPA variant with a mutated receptor docking site, abolished both the effects of uPA on vascular contractility and permeability, without affecting its catalytic activity. These data show that uPA, at concentrations found under pathological conditions, reduces pulmonary arterial contractility and increases permeability though the activation of NMDA-R1. The selective inhibition of NMDAR-1 activation by uPA can be accomplished without a loss of fibrinolytic activity.
Figures

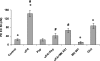
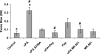

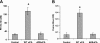
References
-
- Shetty S, Padijnayayveetil J, Tucker T, Stankowska D, Idell S. The fibrinolytic system and the regulation of lung epithelial cell proteolysis, signaling, and cellular viability. Am J Physiol Lung Cell Mol Physiol 2008;295:L967–L975 - PubMed
-
- Idell S. Coagulation, fibrinolysis, and fibrin deposition in acute lung injury. Crit Care Med 2003;31:S213–S220 - PubMed
-
- Bertozzi P, Astedt B, Zenzius L, Lynch K, LeMaire F, Zapol W, Chapman H. Depressed bronchoalveolar urokinase activity in patients with adult respiratory distress syndrome. N Engl J Med 1990;322:890–897 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

