Proteomics identification of ITGB3 as a key regulator in reactive oxygen species-induced migration and invasion of colorectal cancer cells
- PMID: 21622897
- PMCID: PMC3205852
- DOI: 10.1074/mcp.M110.005397
Proteomics identification of ITGB3 as a key regulator in reactive oxygen species-induced migration and invasion of colorectal cancer cells
Abstract
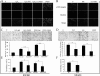
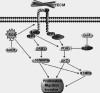
Colorectal cancer (CRC) is the third most commonly diagnosed cancer in males and second in females worldwide. Unfortunately 40-50% of patients already have metastatic disease at presentation when prognosis is poor with a 5-year survival of <10%. Reactive oxygen species (ROS) have been proposed to play a crucial role in tumor metastasis. We now show that higher levels of ROS accumulation are found in a colorectal cancer-derived metastatic cell line (SW620) compared with a cell line (SW480) derived from the primary lesion from the same patient. In addition, ROS accumulation can affect both the migratory and invasive capacity of SW480 and SW620 cells. To explore the molecular mechanism underlying ROS-induced migration and invasion in CRC, we have compared protein expression patterns between SW480 and SW620 cells using a two-dimensional electrophoresis-based proteomics strategy. A total of 63 altered proteins were identified from tandem MS analysis. Cluster analysis revealed dysregulated expression of multiple redox regulative or ROS responsive proteins, implicating their functional roles in colorectal cancer metastasis. Molecular and pathological validation demonstrated that altered expression of PGAM1, GRB2, DJ-1, ITGB3, SOD-1, and STMN1 was closely correlated with the metastatic potential of CRC. Functional studies showed that ROS markedly up-regulated expression of ITGB3, which in turn promoted an aggressive phenotype in SW480 cells, with concomitant up-regulated expression of STMN1. In contrast, knockdown of ITGB3 expression could mitigate the migratory and invasive potential of SW620 or H(2)O(2)-treated SW480 cells, accompanied by down-regulated expression of STMN1. The function of ITGB3 was dependent on the surface expression of integrin αvβ3 heterodimer. Furthermore, STMN1 expression and the PI3K-Akt-mTOR pathway were found to be involved in ROS-induced and ITGB3-mediated migration and invasion of colorectal cancer cells. Taken together, these studies suggest that ITGB3 plays an important role in ROS-induced migration and invasion in CRC.
Figures








Similar articles
-
CXCL12-mediated HOXB5 overexpression facilitates Colorectal Cancer metastasis through transactivating CXCR4 and ITGB3.Theranostics. 2021 Jan 1;11(6):2612-2633. doi: 10.7150/thno.52199. eCollection 2021. Theranostics. 2021. PMID: 33456563 Free PMC article.
-
Extracellular nicotinamide phosphoribosyltransferase visfatin activates JAK2-STAT3 pathway in cancer-associated fibroblasts to promote colorectal cancer metastasis.Genes Genomics. 2025 May;47(5):615-624. doi: 10.1007/s13258-024-01596-6. Epub 2024 Dec 6. Genes Genomics. 2025. PMID: 39643827 Free PMC article.
-
Nuclear lncRNA HOXD-AS1 suppresses colorectal carcinoma growth and metastasis via inhibiting HOXD3-induced integrin β3 transcriptional activating and MAPK/AKT signalling.Mol Cancer. 2019 Mar 1;18(1):31. doi: 10.1186/s12943-019-0955-9. Mol Cancer. 2019. PMID: 30823921 Free PMC article.
-
Redox regulation of cell migration and adhesion.Trends Cell Biol. 2012 Feb;22(2):107-15. doi: 10.1016/j.tcb.2011.11.002. Epub 2011 Dec 31. Trends Cell Biol. 2012. PMID: 22209517 Free PMC article. Review.
-
The Role of Reactive Oxygen Species in Colorectal Cancer Initiation and Progression: Perspectives on Theranostic Approaches.Cancers (Basel). 2025 Feb 22;17(5):752. doi: 10.3390/cancers17050752. Cancers (Basel). 2025. PMID: 40075600 Free PMC article. Review.
Cited by
-
Metastatic suppression by DOC2B is mediated by inhibition of epithelial-mesenchymal transition and induction of senescence.Cell Biol Toxicol. 2022 Apr;38(2):237-258. doi: 10.1007/s10565-021-09598-w. Epub 2021 Mar 24. Cell Biol Toxicol. 2022. PMID: 33758996 Free PMC article.
-
Integrative multi-platform meta-analysis of gene expression profiles in pancreatic ductal adenocarcinoma patients for identifying novel diagnostic biomarkers.PLoS One. 2018 Apr 4;13(4):e0194844. doi: 10.1371/journal.pone.0194844. eCollection 2018. PLoS One. 2018. PMID: 29617451 Free PMC article.
-
ZNF32 protects against oxidative stress-induced apoptosis by modulating C1QBP transcription.Oncotarget. 2015 Nov 10;6(35):38107-26. doi: 10.18632/oncotarget.5646. Oncotarget. 2015. PMID: 26497555 Free PMC article.
-
Preclinical In Vitro Model to Assess the Changes in Permeability and Cytotoxicity of Polarized Intestinal Epithelial Cells during Exposure Mimicking Oral or Intravenous Routes: An Example of Arsenite Exposure.Int J Mol Sci. 2022 Apr 27;23(9):4851. doi: 10.3390/ijms23094851. Int J Mol Sci. 2022. PMID: 35563241 Free PMC article.
-
Spatial Proteomic Analysis of Isogenic Metastatic Colorectal Cancer Cells Reveals Key Dysregulated Proteins Associated with Lymph Node, Liver, and Lung Metastasis.Cells. 2022 Jan 27;11(3):447. doi: 10.3390/cells11030447. Cells. 2022. PMID: 35159257 Free PMC article.
References
-
- Saif M. W. (2006) Targeted agents for adjuvant therapy of colon cancer. Clin. Colorectal Canc. 6, 46–51 - PubMed
-
- Etzioni R., Urban N., Ramsey S., McIntosh M., Schwartz S., Reid B., Radich J., Anderson G., Hartwell L. (2003) The case for early detection. Nat. Rev. Cancer 3, 243–252 - PubMed
-
- Chambers A. F., Groom A. C., MacDonald I. C. (2002) Dissemination and growth of cancer cells in metastatic sites. Nat. Rev. Cancer 2, 563–572 - PubMed
-
- Davies R. J., Miller R., Coleman N. (2005) Colorectal cancer screening: prospects for molecular stool analysis. Nat. Rev. Cancer 5, 199–209 - PubMed
-
- Poli G., Leonarduzzi G., Biasi F., Chiarpotto E. (2004) Oxidative stress and cell signalling. Curr. Med. Chem. 11, 1163–1182 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

