Connexin43 interacts with βarrestin: a pre-requisite for osteoblast survival induced by parathyroid hormone
- PMID: 21630325
- PMCID: PMC3178733
- DOI: 10.1002/jcb.23208
Connexin43 interacts with βarrestin: a pre-requisite for osteoblast survival induced by parathyroid hormone
Abstract
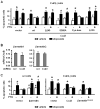
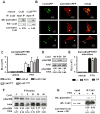
Parathyroid hormone (PTH) promotes osteoblast survival through a mechanism that depends on cAMP-mediated signaling downstream of the G protein-coupled receptor PTHR1. We present evidence herein that PTH-induced survival signaling is impaired in cells lacking connexin43 (Cx43). Thus, expression of functional Cx43 dominant negative proteins or Cx43 knock-down abolished the expression of cAMP-target genes and anti-apoptosis induced by PTH in osteoblastic cells. In contrast, cells lacking Cx43 were still responsive to the stable cAMP analog dibutyril-cAMP. PTH survival signaling was rescued by transfecting wild type Cx43 or a truncated dominant negative mutant of βarrestin, a PTHR1-interacting molecule that limits cAMP signaling. On the other hand, Cx43 mutants lacking the cytoplasmic domain (Cx43(Δ245)) or unable to be phosphorylated at serine 368 (Cx43(S368A)), a residue crucial for Cx43 trafficking and function, failed to restore the anti-apoptotic effect of PTH in Cx43-deficient cells. In addition, overexpression of wild type βarrestin abrogated PTH survival signaling in Cx43-expressing cells. Moreover, βarrestin physically associated in vivo to wild type Cx43 and to a lesser extent to Cx43(S368A) ; and this association and the phosphorylation of Cx43 in serine 368 were reduced by PTH. Furthermore, induction of Cx43(S368) phosphorylation or overexpression of wild type Cx43, but not Cx43(Δ245) or Cx43(S368A) , reduced the interaction between βarrestin and the PTHR1. These studies demonstrate that βarrestin is a novel Cx43-interacting protein and suggest that, by sequestering βarrestin, Cx43 facilitates cAMP signaling, thereby exerting a permissive role on osteoblast survival induced by PTH.
Copyright © 2011 Wiley-Liss, Inc.
Figures


References
-
- Ai X, Pogwizd SM. Connexin 43 downregulation and dephosphorylation in nonischemic heart failure is associated with enhanced colocalized protein phosphatase type 2A. Circ Res. 2005;96:54–63. - PubMed
-
- Attramadal H, Arriza JL, Aoki C, Dawson TM, Codina J, Kwatra MM, Snyder SH, Caron MG, Lefkowitz RJ. Beta-arrestin2, a novel member of the arrestin/beta-arrestin gene family. J Biol Chem. 1992;267:17882–17890. - PubMed
-
- Bao X, Reuss L, Altenberg GA. Regulation of purified and reconstituted connexin 43 hemichannels by protein kinase C-mediated phosphorylation of Serine 368. J Biol Chem. 2004;279:20058–20066. - PubMed
-
- Bellido T, Ali AA, Plotkin LI, Fu Q, Gubrij I, Roberson PK, Weinstein RS, O’Brien CA, Manolagas SC, Jilka RL. Proteasomal degradation of Runx2 shortens parathyroid hormone-induced anti-apoptotic signaling in osteoblasts. A putative explanation for why intermittent administration is needed for bone anabolism. J Biol Chem. 2003;278:50259–50272. - PubMed
-
- Beyer EC, Paul DL, Goodenough DA. Connexin family of gap junction proteins. J Membr Biol. 1990;116:187–194. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

