Real-time snapshot hyperspectral imaging endoscope
- PMID: 21639573
- PMCID: PMC3107836
- DOI: 10.1117/1.3574756
Real-time snapshot hyperspectral imaging endoscope
Abstract
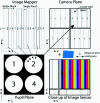
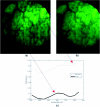
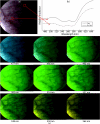
Hyperspectral imaging has tremendous potential to detect important molecular biomarkers of early cancer based on their unique spectral signatures. Several drawbacks have limited its use for in vivo screening applications: most notably the poor temporal and spatial resolution, high expense, and low optical throughput of existing hyperspectral imagers. We present the development of a new real-time hyperspectral endoscope (called the image mapping spectroscopy endoscope) based on an image mapping technique capable of addressing these challenges. The parallel high throughput nature of this technique enables the device to operate at frame rates of 5.2 frames per second while collecting a (x, y, λ) datacube of 350 × 350 × 48. We have successfully imaged tissue in vivo, resolving a vasculature pattern of the lower lip while simultaneously detecting oxy-hemoglobin.
Figures













References
-
- Horner M. J., Ries L. A. G., Krapcho M., Neyman N., Aminou R., Howlader N., Altekruse S. F., Feuer E. J., Huang L., Mariotto A., Miller B. A., Lewis D. R., Eisner M. P., Stinchcomb D. G., and Edwards B. K., Eds., SEER Cancer Statistics Review, 1975–2006, National Cancer Institute, Bethesda, Maryland, http://seer.cancer.gov/csr/1975_2006/, based on November 2008 SEER data submission, posted to the SEER web site, 2009.
-
- Drezek R., Brookner C., Pavlova I., Boiko I., Malpica A., Lotan R., Follen M., and Richards-Kortum R., “Autofluorescence microscopy of fresh cervical-tissue sections reveals alterations in tissue biochemistry with dysplasia,” Photochem. Photobiol. 73, 636–641 (2001).10.1562/0031-8655(2001)0730636AMOFCT2.0.CO2 - DOI - PubMed
-
- Wu Y., Zheng W., and Qu J. Y., “Time-resolved confocal fluorescence spectroscopy reveals the structure and metabolic state of epithelial tissue,” Proc. SPIE 6430, 643013 (2007).10.1117/12.702450 - DOI
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

