Stage-specific changes in GDNF expression by rat Sertoli cells: a possible regulator of the replication and differentiation of stem spermatogonia
- PMID: 21653894
- PMCID: PMC3184291
- DOI: 10.1095/biolreprod.110.087676
Stage-specific changes in GDNF expression by rat Sertoli cells: a possible regulator of the replication and differentiation of stem spermatogonia
Abstract
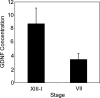
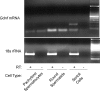
In the adult testis, the precise control of the self-renewing replication and differentiation of stem spermatogonia is fundamental to male fertility. Previous studies have shown that the replication of A single (A(s)) spermatogonia, a population that includes the stem cells, is maximal at stage I of the cycle of the rat seminiferous epithelium and minimal at stage VII, while the ratio of A-paired spermatogonia to A(s) spermatogonia increases from stages I to VII. It has been hypothesized that these changes in A(s) spermatogonia replication and differentiation result from changes in the expression of glial cell-line derived neurotrophic factor (GDNF) by Sertoli cells. To directly test this hypothesis, we used immunocytochemistry and confocal microscopy to demonstrate that within intact seminiferous tubules, GDNF is detectable only in Sertoli cells and that its amount and its location within these cells changes with progression of the stages of the cycle. The identification of Sertoli cells as the primary source of GDNF was confirmed by RT-PCR analysis of RNA isolated from purified populations of Sertoli cells, pachytene spermatocytes, and round spermatids. Stage-specific changes in GDNF expression were confirmed by quantifying GDNF mRNA in seminiferous tubules at defined stages of the cycle. Expression of this transcript was maximal at stage I, fell 14-fold by stage VIIc,d, and then increased 12-fold by stages XIII-XIV. This pattern of expression was the opposite of the control, cathepsin L mRNA. Taken together, these data support the hypothesis that cyclical changes in GDNF expression by Sertoli cells are responsible for the stage-specific replication and differentiation of stem spermatogonia, the foundational cells of spermatogenesis.
Figures


References
-
- Huckins C. The spermatogonial stem cell population in adult rats. I. Their morphology, proliferation and maturation. Anat Rec 1971; 169: 533 557 - PubMed
-
- Orwig KE, Shinohara T, Avarbock MR, Brinster RL. Functional analysis of stem cells in the adult rat testis. Biol Reprod 2002; 66: 944 949 - PubMed
-
- Wing TY, Christensen AK. Morphometric studies on rat seminiferous tubules. Am J Anat 1982; 165: 13 25 - PubMed
-
- Huckins C. The spermatogonial stem cell population in adult rats. II. A radioautographic analysis of their cell cycle properties. Cell Tissue Kinet 1971; 4: 313 334 - PubMed
-
- Huckins C. The spermatogonial stem cell population in adult rats. 3. Evidence for a long-cycling population. Cell Tissue Kinet 1971; 4: 335 349 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

