Two-dimensional intravascular near-infrared fluorescence molecular imaging of inflammation in atherosclerosis and stent-induced vascular injury
- PMID: 21679853
- PMCID: PMC3123768
- DOI: 10.1016/j.jacc.2011.02.036
Two-dimensional intravascular near-infrared fluorescence molecular imaging of inflammation in atherosclerosis and stent-induced vascular injury
Abstract
Objectives: This study sought to develop a 2-dimensional (2D) intravascular near-infrared fluorescence (NIRF) imaging strategy for investigation of arterial inflammation in coronary-sized vessels.
Background: Molecular imaging of arterial inflammation could provide new insights into the pathogenesis of acute myocardial infarction stemming from coronary atheromata and implanted stents. Presently, few high-resolution approaches can image inflammation in coronary-sized arteries in vivo.
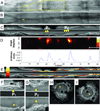
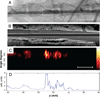
Methods: A new 2.9-F rotational, automated pullback 2D imaging catheter was engineered and optimized for 360° viewing intravascular NIRF imaging. In conjunction with the cysteine protease-activatable imaging reporter Prosense VM110 (VisEn Medical, Woburn, Massachusetts), intra-arterial 2D NIRF imaging was performed in rabbit aortas with atherosclerosis (n =10) or implanted coronary bare-metal stents (n = 10, 3.5-mm diameter, day 7 post-implantation). Intravascular ultrasound provided coregistered anatomical images of arteries. After sacrifice, specimens underwent ex vivo NIRF imaging, fluorescence microscopy, and histological and immunohistochemical analyses.
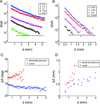
Results: Imaging of coronary artery-scaled phantoms demonstrated 8-sector angular resolution and submillimeter axial resolution, nanomolar sensitivity to NIR fluorochromes, and modest NIRF light attenuation through blood. High-resolution NIRF images of vessel wall inflammation with signal-to-noise ratios >10 were obtained in real-time through blood, without flushing or occlusion. In atherosclerosis, 2D NIRF, intravascular ultrasound-NIRF fusion, microscopy, and immunoblotting studies provided insight into the spatial distribution of plaque protease activity. In stent-implanted vessels, real-time imaging illuminated an edge-based pattern of stent-induced arterial inflammation.
Conclusions: A new 2D intravascular NIRF imaging strategy provides high-resolution in vivo spatial mapping of arterial inflammation in coronary-sized arteries and reveals increased inflammation-regulated cysteine protease activity in atheromata and stent-induced arterial injury.
Copyright © 2011 American College of Cardiology Foundation. Published by Elsevier Inc. All rights reserved.
Figures








References
-
- Libby P. Inflammation in atherosclerosis. Nature. 2002;420:868–874. - PubMed
-
- Rader DJ, Daugherty A. Translating molecular discoveries into new therapies for atherosclerosis. Nature. 2008;451:904–913. - PubMed
-
- Luscher TF, Steffel J, Eberli FR, et al. Drug-eluting stent and coronary thrombosis: biological mechanisms and clinical implications. Circulation. 2007;115:1051–1058. - PubMed
-
- Nakazawa G, Finn AV, Joner M, et al. Delayed arterial healing and increased late stent thrombosis at culprit sites after drug-eluting stent placement for acute myocardial infarction patients: an autopsy study. Circulation. 2008;118:1138–1145. - PubMed
-
- Jaffer FA, Libby P, Weissleder R. Molecular imaging of cardiovascular disease. Circulation. 2007;116:1052–1061. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

