Growth of limb muscle is dependent on skeletal-derived Indian hedgehog
- PMID: 21683695
- PMCID: PMC3322465
- DOI: 10.1016/j.ydbio.2011.06.002
Growth of limb muscle is dependent on skeletal-derived Indian hedgehog
Abstract
During embryogenesis, muscle and bone develop in close temporal and spatial proximity. We show that Indian Hedgehog, a bone-derived signaling molecule, participates in growth of skeletal muscle. In Ihh(-/-) embryos, skeletal muscle development appears abnormal at embryonic day 14.5 and at later ages through embryonic day 20.5, dramatic losses of hindlimb muscle occur. To further examine the role of Ihh in myogenesis, we manipulated Ihh expression in the developing chick hindlimb. Reduction of Ihh in chicken embryo hindlimbs reduced skeletal muscle mass similar to that seen in Ihh(-/-) mouse embryos. The reduction in muscle mass appears to be a direct effect of Ihh since ectopic expression of Ihh by RCAS retroviral infection of chicken embryo hindlimbs restores muscle mass. These effects are independent of bone length, and occur when Shh is not expressed, suggesting Ihh acts directly on fetal myoblasts to regulate secondary myogenesis. Loss of muscle mass in Ihh null mouse embryos is accompanied by a dramatic increase in myoblast apoptosis by a loss of p21 protein. Our data suggest that Ihh promotes fetal myoblast survival during their differentiation into secondary myofibers by maintaining p21 protein levels.
Copyright © 2011 Elsevier Inc. All rights reserved.
Figures
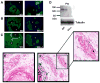
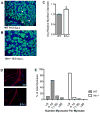


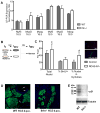
References
-
- Ahlgren SC, Bronner-Fraser M. Inhibition of sonic hedgehog signaling in vivo results in craniofacial neural crest cell death. Curr Biol. 1999;9:1304–1314. - PubMed
-
- Ayers KL, et al. The long-range activity of Hedgehog is regulated in the apical extracellular space by the glypican Dally and the hydrolase Notum. Dev Cell. 2010;18:605–620. - PubMed
-
- Biressi S, et al. Cellular heterogeneity during vertebrate skeletal muscle development. Dev Biol. 2007;308:281–293. - PubMed
-
- Biressi S, et al. Intrinsic phenotypic diversity of embryonic and fetal myoblasts is revealed by genome-wide gene expression analysis on purified cells. Dev Biol. 2007;304:633–651. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

