Fibrinogen β-derived Bβ(15-42) peptide protects against kidney ischemia/reperfusion injury
- PMID: 21685370
- PMCID: PMC3158721
- DOI: 10.1182/blood-2011-02-338061
Fibrinogen β-derived Bβ(15-42) peptide protects against kidney ischemia/reperfusion injury
Abstract
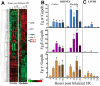
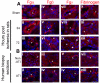
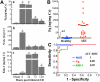
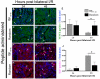
Ischemia/reperfusion (I/R) injury in the kidney is a major cause of acute kidney injury (AKI) in humans and is associated with significantly high mortality. To identify genes that modulate kidney injury and repair, we conducted genome-wide expression analysis in the rat kidneys after I/R and found that the mRNA levels of fibrinogen (Fg)α, Fgβ, and Fgγ chains significantly increase in the kidney and remain elevated throughout the regeneration process. Cellular characterization of Fgα and Fgγ chain immunoreactive proteins shows a predominant expression in renal tubular cells and the localization of immunoreactive Fgβ chain protein is primarily in the renal interstitium in healthy and regenerating kidney. We also show that urinary excretion of Fg is massively increased after kidney damage and is capable of distinguishing human patients with acute or chronic kidney injury (n = 25) from healthy volunteers (n = 25) with high sensitivity and specificity (area under the receiver operating characteristic of 0.98). Furthermore, we demonstrate that Fgβ-derived Bβ(15-42) peptide administration protects mice from I/R-induced kidney injury by aiding in epithelial cell proliferation and tissue repair. Given that kidney regeneration is a major determinant of outcome for patients with kidney damage, these results provide new opportunities for the use of Fg in diagnosis, prevention, and therapeutic interventions in kidney disease.
Figures

References
-
- Eckardt KU, Kasiske BL. Kidney disease: improving global outcomes. Nat Rev Nephrol. 2009;5(11):650–657. - PubMed
-
- Szczech LA, Harmon W, Hostetter TH, et al. World Kidney Day 2009: problems and challenges in the emerging epidemic of kidney disease. J Am Soc Nephrol. 2009;20(3):453–455. - PubMed
-
- Basile DP. Novel approaches in the investigation of acute kidney injury. J Am Soc Nephrol. 2007;18(1):7–9. - PubMed
-
- Reinders ME, Rabelink TJ, Briscoe DM. Angiogenesis and endothelial cell repair in renal disease and allograft rejection. J Am Soc Nephrol. 2006;17(4):932–942. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

