Adipocyte enhancer-binding protein 1 (AEBP1) (a novel macrophage proinflammatory mediator) overexpression promotes and ablation attenuates atherosclerosis in ApoE (-/-) and LDLR (-/-) mice
- PMID: 21687917
- PMCID: PMC3188858
- DOI: 10.2119/molmed.2011.00141
Adipocyte enhancer-binding protein 1 (AEBP1) (a novel macrophage proinflammatory mediator) overexpression promotes and ablation attenuates atherosclerosis in ApoE (-/-) and LDLR (-/-) mice
Abstract
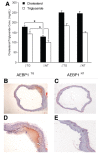
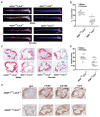
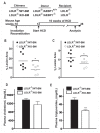
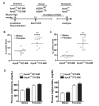
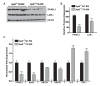
Atherogenesis is a long-term process that involves inflammatory response coupled with metabolic dysfunction. Foam cell formation and macrophage inflammatory response are two key events in atherogenesis. Adipocyte enhancer-binding protein 1 (AEBP1) has been shown to impede macrophage cholesterol efflux, promoting foam cell formation, via peroxisome proliferator-activated receptor (PPAR)-γ1 and liver X receptor α (LXRα) downregulation. Moreover, AEBP1 has been shown to promote macrophage inflammatory responsiveness by inducing nuclear factor (NF)-κB activity via IκBα downregulation. Lipopolysaccharide (LPS)-induced suppression of pivotal macrophage cholesterol efflux mediators, leading to foam cell formation, has been shown to be mediated by AEBP1. Herein, we showed that AEBP1-transgenic mice (AEBP1(TG)) with macrophage-specific AEBP1 overexpression exhibit hyperlipidemia and develop atherosclerotic lesions in their proximal aortas. Consistently, ablation of AEBP1 results in significant attenuation of atherosclerosis (males: 3.2-fold, P = 0.001 [en face]), 2.7-fold, P = 0.0004 [aortic roots]; females: 2.1-fold, P = 0.0026 [en face], 1.7-fold, P = 0.0126 [aortic roots]) in the AEBP1(-/-)/low-density lipoprotein receptor (LDLR )(-/-) double-knockout (KO) mice. Bone marrow (BM) transplantation experiments further revealed that LDLR (-/-) mice reconstituted with AEBP1(-/-)/LDLR (-/-) BM cells (LDLR (-/-)/KO-BM chimera) display significant reduction of atherosclerosis lesions (en face: 2.0-fold, P = 0.0268; aortic roots: 1.7-fold, P = 0.05) compared with control mice reconstituted with AEBP1(+/+)/LDLR (-/-) BM cells (LDLR (-/-)/WT-BM chimera). Furthermore, transplantation of AEBP1(TG) BM cells with the normal apolipoprotein E (ApoE) gene into ApoE (-/-) mice (ApoE (-/-)/TG-BM chimera) leads to significant development of atherosclerosis (males: 2.5-fold, P = 0.0001 [en face], 4.7-fold, P = 0.0001 [aortic roots]; females: 1.8-fold, P = 0.0001 [en face], 3.0-fold, P = 0.0001 [aortic roots]) despite the restoration of ApoE expression. Macrophages from ApoE (-/-)/TG-BM chimeric mice express reduced levels of PPARγ1, LXRα, ATP-binding cassette A1 (ABCA1) and ATP-binding cassette G1 (ABCG1) and increased levels of the inflammatory mediators interleukin (IL)-6 and tumor necrosis factor (TNF)-α compared with macrophages of control chimeric mice (ApoE (-/-)/NT-BM ) that received AEBP1 nontransgenic (AEBP1(NT) ) BM cells. Our in vivo experimental data strongly suggest that macrophage AEBP1 plays critical regulatory roles in atherogenesis, and it may serve as a potential therapeutic target for the prevention or treatment of atherosclerosis.
Figures

References
-
- Ross R. Atherosclerosis: an inflammatory disease. N Engl J Med. 1999;340:115–26. - PubMed
-
- Libby P. Inflammation in atherosclerosis. Nature. 2002;420:868–74. - PubMed
-
- Tontonoz P, Nagy L, Alvarez JG, Thomazy VA, Evans RM. PPARgamma promotes monocyte/macrophage differentiation and uptake of oxidized LDL. Cell. 1998;93:241–52. - PubMed
-
- Nagy L, Tontonoz P, Alvarez JG, Chen H, Evans RM. Oxidized LDL regulates macrophage gene expression through ligand activation of PPARgamma. Cell. 1998;93:229–40. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

