Protein kinase D1 (PKD1) activation mediates a compensatory protective response during early stages of oxidative stress-induced neuronal degeneration
- PMID: 21696630
- PMCID: PMC3145571
- DOI: 10.1186/1750-1326-6-43
Protein kinase D1 (PKD1) activation mediates a compensatory protective response during early stages of oxidative stress-induced neuronal degeneration
Abstract
Background: Oxidative stress is a key pathophysiological mechanism contributing to degenerative processes in many neurodegenerative diseases and therefore, unraveling molecular mechanisms underlying various stages of oxidative neuronal damage is critical to better understanding the diseases and developing new treatment modalities. We previously showed that protein kinase C delta (PKCδ) proteolytic activation during the late stages of oxidative stress is a key proapoptotic signaling mechanism that contributes to oxidative damage in Parkinson's disease (PD) models. The time course studies revealed that PKCδ activation precedes apoptotic cell death and that cells resisted early insults of oxidative damage, suggesting that some intrinsic compensatory response protects neurons from early oxidative insult. Therefore, the purpose of the present study was to characterize protective signaling pathways in dopaminergic neurons during early stages of oxidative stress.
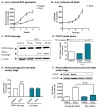
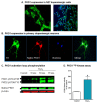
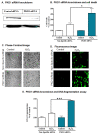
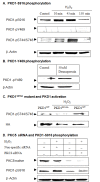
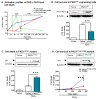
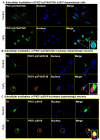
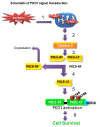
Results: Herein, we identify that protein kinase D1 (PKD1) functions as a key anti-apoptotic kinase to protect neuronal cells against early stages of oxidative stress. Exposure of dopaminergic neuronal cells to H2O2 or 6-OHDA induced PKD1 activation loop (PKD1S744/748) phosphorylation long before induction of neuronal cell death. Blockade of PKCδ cleavage, PKCδ knockdown or overexpression of a cleavage-resistant PKCδ mutant effectively attenuated PKD1 activation, indicating that PKCδ proteolytic activation regulates PKD1 phosphorylation. Furthermore, the PKCδ catalytic fragment, but not the regulatory fragment, increased PKD1 activation, confirming PKCδ activity modulates PKD1 activation. We also identified that phosphorylation of S916 at the C-terminal is a preceding event required for PKD1 activation loop phosphorylation. Importantly, negative modulation of PKD1 by the RNAi knockdown or overexpression of PKD1S916A phospho-defective mutants augmented oxidative stress-induced apoptosis, while positive modulation of PKD1 by the overexpression of full length PKD1 or constitutively active PKD1 plasmids attenuated oxidative stress-induced apoptosis, suggesting an anti-apoptotic role for PKD1 during oxidative neuronal injury.
Conclusion: Collectively, our results demonstrate that PKCδ-dependent activation of PKD1 represents a novel intrinsic protective response in counteracting early stage oxidative damage in neuronal cells. Our results suggest that positive modulation of the PKD1-mediated compensatory protective mechanism against oxidative damage in dopaminergic neurons may provide novel neuroprotective strategies for treatment of PD.
Figures



Similar articles
-
Protein kinase D1 (PKD1) phosphorylation promotes dopaminergic neuronal survival during 6-OHDA-induced oxidative stress.PLoS One. 2014 May 7;9(5):e96947. doi: 10.1371/journal.pone.0096947. eCollection 2014. PLoS One. 2014. PMID: 24806360 Free PMC article.
-
Dopaminergic neurotoxicant 6-OHDA induces oxidative damage through proteolytic activation of PKCδ in cell culture and animal models of Parkinson's disease.Toxicol Appl Pharmacol. 2011 Nov 1;256(3):314-23. doi: 10.1016/j.taap.2011.07.021. Epub 2011 Aug 6. Toxicol Appl Pharmacol. 2011. PMID: 21846476 Free PMC article.
-
Environmental neurotoxic pesticide dieldrin activates a non receptor tyrosine kinase to promote PKCδ-mediated dopaminergic apoptosis in a dopaminergic neuronal cell model.Neurotoxicology. 2011 Oct;32(5):567-77. doi: 10.1016/j.neuro.2011.06.009. Epub 2011 Jul 23. Neurotoxicology. 2011. PMID: 21801747 Free PMC article.
-
Role of proteolytic activation of protein kinase Cdelta in oxidative stress-induced apoptosis.Antioxid Redox Signal. 2003 Oct;5(5):609-20. doi: 10.1089/152308603770310275. Antioxid Redox Signal. 2003. PMID: 14580317 Review.
-
Role of protein kinase Cδ in dopaminergic neurotoxic events.Food Chem Toxicol. 2018 Nov;121:254-261. doi: 10.1016/j.fct.2018.09.005. Epub 2018 Sep 6. Food Chem Toxicol. 2018. PMID: 30195712 Review.
Cited by
-
Methamphetamine-induced neurotoxicity linked to ubiquitin-proteasome system dysfunction and autophagy-related changes that can be modulated by protein kinase C delta in dopaminergic neuronal cells.Neuroscience. 2012 May 17;210:308-32. doi: 10.1016/j.neuroscience.2012.03.004. Epub 2012 Mar 14. Neuroscience. 2012. PMID: 22445524 Free PMC article.
-
Protein kinase D1 (PKD1) phosphorylation promotes dopaminergic neuronal survival during 6-OHDA-induced oxidative stress.PLoS One. 2014 May 7;9(5):e96947. doi: 10.1371/journal.pone.0096947. eCollection 2014. PLoS One. 2014. PMID: 24806360 Free PMC article.
-
Molecular mechanisms underlying protective effects of quercetin against mitochondrial dysfunction and progressive dopaminergic neurodegeneration in cell culture and MitoPark transgenic mouse models of Parkinson's Disease.J Neurochem. 2017 Jun;141(5):766-782. doi: 10.1111/jnc.14033. Epub 2017 May 9. J Neurochem. 2017. PMID: 28376279 Free PMC article.
-
Single-Cell Landscape of the Cochlea Revealed Cell-Type-Specific Diversification in Hipposideros armiger Based on PacBio Long-Read Sequencing.Biomolecules. 2025 Feb 1;15(2):211. doi: 10.3390/biom15020211. Biomolecules. 2025. PMID: 40001514 Free PMC article.
-
Oxidant stress and signal transduction in the nervous system with the PI 3-K, Akt, and mTOR cascade.Int J Mol Sci. 2012 Oct 26;13(11):13830-66. doi: 10.3390/ijms131113830. Int J Mol Sci. 2012. PMID: 23203037 Free PMC article. Review.
References
-
- Kanthasamy A, Kitazawa M, Kaul S, Anantharam V, Kanthasamy AG. A novel oxidative stress-dependent apoptotic pathway in pesticide-induced dopaminergic degeneration: relevance to environmental factors and Parkinson's disease. J Neurochem. 2002;81(suppl 1):76.
-
- Przedborski S. Pathogenesis of nigral cell death in Parkinson's disease. Parkinsonism Relat Disord. 2005;11(Suppl 1):S3–7. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

