Cholecalciferol plus calcium suppresses abnormal PBMC reactivity in patients with multiple sclerosis
- PMID: 21697250
- PMCID: PMC3417163
- DOI: 10.1210/jc.2011-0325
Cholecalciferol plus calcium suppresses abnormal PBMC reactivity in patients with multiple sclerosis
Abstract
Context: The active metabolite of vitamin D, 1,25-dihydroxyvitamin D [1,25(OH)(2)D], is a potent modulator of immune cells in vitro.
Objective: Our objective was to determine whether the sun-dependent nutrient, cholecalciferol, can alter disease-associated cellular immune abnormalities in patients with multiple sclerosis (MS).
Design: This was an open-label, 12-month, randomized controlled trial.
Setting: Patients with MS were recruited from the MS Clinic at St. Michael's Hospital, Toronto.
Patients: Forty-nine patients were matched (for age, sex, disease duration, disease-modifying drug, and disability) and enrolled (treated n = 25; control n = 24). Four patients were lost to follow-up (n = 2 from each group).
Intervention: Treated patients received increasing doses of cholecalciferol (4,000-40,000 IU/d) plus calcium (1200 mg/d), followed by equilibration to a moderate, physiological intake (10,000 IU/d). Control patients did not receive supplements.
Main outcome measures: At enrollment and at 12 months, peripheral blood mononuclear cell (PBMC) proliferative responses to disease-associated, MS-relevant, and control antigens were measured, along with selected serum biochemical markers.
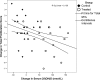
Results: At 12 months, mean serum 25-hydroxyvitamin D [25(OH)D] concentrations were 83 ± 35 nmol/liter and 179 ± 76 nmol/liter in control and treated participants, respectively (paired t, P < 0.001). Serum 1,25(OH)(2)D did not differ between baseline and 1 yr. In treated patients, 12-month PBMC proliferative responses to neuron antigens myelin basic protein and exon-2 were suppressed (P = 0.002). In controls, there were no significant changes in disease-associated PBMC responsiveness. There were no significant differences between groups in levels of selected biomarkers.
Interpretation: MS-associated, abnormal T cell reactivities were suppressed in vivo by cholecalciferol at serum 25(OH)D concentrations higher than 100 nmol/liter.
Trial registration: ClinicalTrials.gov NCT00644904.
Figures

 (blood drop, indicating blood collection).
(blood drop, indicating blood collection).
References
-
- Winer S, Astsaturov I, Cheung R, Gunaratnam L, Kubiak V, Cortez MA, Moscarello M, O'Connor PW, McKerlie C, Becker DJ, Dosch HM. 2001. Type I diabetes and multiple sclerosis patients target islet plus central nervous system autoantigens: nonimmunized nonobese diabetic mice can develop autoimmune encephalitis. J Immunol 166:2831–2841 - PubMed
-
- Banwell B, Bar-Or A, Cheung R, Kennedy J, Krupp LB, Becker DJ, Dosch HM. 2008. Abnormal T-cell reactivities in childhood inflammatory demyelinating disease and type 1 diabetes. Ann Neurol 63:98–111 - PubMed
-
- Steinman L. 2004. Immune therapy for autoimmune diseases. Science 305:212–216 - PubMed
-
- Vieth R. 1999. Vitamin D supplementation, 25-hydroxyvitamin D concentrations, and safety. Am J Clin Nutr 69:842–856 - PubMed

