MicroRNA regulation of homeostatic synaptic plasticity
- PMID: 21697510
- PMCID: PMC3136313
- DOI: 10.1073/pnas.1017576108
MicroRNA regulation of homeostatic synaptic plasticity
Abstract
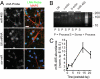
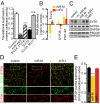
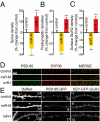
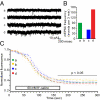
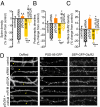
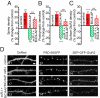
Homeostatic mechanisms are required to control formation and maintenance of synaptic connections to maintain the general level of neural impulse activity within normal limits. How genes controlling these processes are co-coordinately regulated during homeostatic synaptic plasticity is unknown. MicroRNAs (miRNAs) exert regulatory control over mRNA stability and translation and may contribute to local and activity-dependent posttranscriptional control of synapse-associated mRNAs. However, identifying miRNAs that function through posttranscriptional gene silencing at synapses has remained elusive. Using a bioinformatics screen to identify sequence motifs enriched in the 3'UTR of rapidly destabilized mRNAs, we identified a developmentally and activity-regulated miRNA (miR-485) that controls dendritic spine number and synapse formation in an activity-dependent homeostatic manner. We find that many plasticity-associated genes contain predicted miR-485 binding sites and further identify the presynaptic protein SV2A as a target of miR-485. miR-485 negatively regulated dendritic spine density, postsynaptic density 95 (PSD-95) clustering, and surface expression of GluR2. Furthermore, miR-485 overexpression reduced spontaneous synaptic responses and transmitter release, as measured by miniature excitatory postsynaptic current (EPSC) analysis and FM 1-43 staining. SV2A knockdown mimicked the effects of miR-485, and these effects were reversed by SV2A overexpression. Moreover, 5 d of increased synaptic activity induced homeostatic changes in synaptic specializations that were blocked by a miR-485 inhibitor. Our findings reveal a role for this previously uncharacterized miRNA and the presynaptic protein SV2A in homeostatic plasticity and nervous system development, with possible implications in neurological disorders (e.g., Huntington and Alzheimer's disease), where miR-485 has been found to be dysregulated.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Systematic identification of 3'-UTR regulatory elements in activity-dependent mRNA stability in hippocampal neurons.Philos Trans R Soc Lond B Biol Sci. 2014 Sep 26;369(1652):20130509. doi: 10.1098/rstb.2013.0509. Philos Trans R Soc Lond B Biol Sci. 2014. PMID: 25135970 Free PMC article.
-
MicroRNA regulation of the synaptic plasticity-related gene Arc.PLoS One. 2012;7(7):e41688. doi: 10.1371/journal.pone.0041688. Epub 2012 Jul 26. PLoS One. 2012. PMID: 22844515 Free PMC article.
-
An activity-regulated microRNA, miR-188, controls dendritic plasticity and synaptic transmission by downregulating neuropilin-2.J Neurosci. 2012 Apr 18;32(16):5678-5687. doi: 10.1523/JNEUROSCI.6471-11.2012. J Neurosci. 2012. PMID: 22514329 Free PMC article.
-
Making of a Synapse: Recurrent Roles of Drebrin A at Excitatory Synapses Throughout Life.Adv Exp Med Biol. 2017;1006:119-139. doi: 10.1007/978-4-431-56550-5_8. Adv Exp Med Biol. 2017. PMID: 28865018 Review.
-
Molecular mechanisms that underlie structural and functional changes at the postsynaptic membrane during synaptic plasticity.Prog Neurobiol. 1998 Aug;55(6):611-40. doi: 10.1016/s0301-0082(98)00026-4. Prog Neurobiol. 1998. PMID: 9670221 Review.
Cited by
-
MicroRNAs in the aging female brain: a putative mechanism for age-specific estrogen effects.Endocrinology. 2013 Aug;154(8):2795-806. doi: 10.1210/en.2013-1230. Epub 2013 May 29. Endocrinology. 2013. PMID: 23720423 Free PMC article.
-
Skeletal muscle calpain acts through nitric oxide and neural miRNAs to regulate acetylcholine release in motor nerve terminals.J Neurosci. 2013 Apr 24;33(17):7308-7324. doi: 10.1523/JNEUROSCI.0224-13.2013. J Neurosci. 2013. PMID: 23616539 Free PMC article.
-
MicroRNAs of the miR379-410 cluster: New players in embryonic neurogenesis and regulators of neuronal function.Neurogenesis (Austin). 2015 Mar 4;2(1):e1004970. doi: 10.1080/23262133.2015.1004970. eCollection 2015. Neurogenesis (Austin). 2015. PMID: 27504472 Free PMC article. Review.
-
A placental mammal-specific microRNA cluster acts as a natural brake for sociability in mice.EMBO Rep. 2019 Feb;20(2):e46429. doi: 10.15252/embr.201846429. Epub 2018 Dec 14. EMBO Rep. 2019. PMID: 30552145 Free PMC article.
-
Hippocampal stem cells promotes synaptic resistance to the dysfunctional impact of amyloid beta oligomers via secreted exosomes.Mol Neurodegener. 2019 Jun 14;14(1):25. doi: 10.1186/s13024-019-0322-8. Mol Neurodegener. 2019. PMID: 31200742 Free PMC article.
References
-
- Turrigiano GG, Leslie KR, Desai NS, Rutherford LC, Nelson SB. Activity-dependent scaling of quantal amplitude in neocortical neurons. Nature. 1998;391:892–896. - PubMed
-
- Hebb DO. The Organization of Behavior: A Neuropsychological Theory. New York: Wiley; 1949.
-
- Thiagarajan TC, Lindskog M, Tsien RW. Adaptation to synaptic inactivity in hippocampal neurons. Neuron. 2005;47:725–737. - PubMed
-
- Thiagarajan TC, Piedras-Renteria ES, Tsien RW. alpha- and betaCaMKII. Inverse regulation by neuronal activity and opposing effects on synaptic strength. Neuron. 2002;36:1103–1114. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

