The contribution of vitamin A to autocrine regulation of fat depots
- PMID: 21704731
- PMCID: PMC3196743
- DOI: 10.1016/j.bbalip.2011.06.004
The contribution of vitamin A to autocrine regulation of fat depots
Abstract
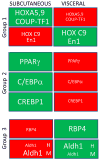
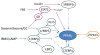
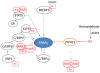
Morbidity and mortality associated with increased white fat accumulation in visceral fat depots have focused attention on the pathways regulating the development of this tissue during embryogenesis, in adulthood, and while under the influence of obesogenic diets. Adipocytes undergo clonal expansion, differentiation (adipogenesis) and maturation through a complex network of transcriptional factors, most of which are expressed at similar levels in visceral and subcutaneous fat. Rigorous research attempts to unfold the pathways regulating expression and activity of adipogenic transcription factors that act in a fat-depot-specific manner. Peroxisome proliferator-activated receptor-γ (PPARγ) is the master regulator of adipogenesis, and is expressed at higher levels in subcutaneous than in visceral depots. PPARγ expression in adipogenesis is mediated by CCAAT/enhancer binding proteins (C/EBPs) and several transcription factors acting in conjunction with C/EBPs, although alternative pathways through zinc-finger protein-423 (ZFP423) transcription factor are sufficient to induce PPARγ expression and adipogenesis. Vitamin A and its metabolites, retinaldehyde and retinoic acid, are transcriptionally-active molecules. Retinoic acid is generated from retinaldehyde in adipose tissue by the aldehyde dehydrogenase-1 family of enzymes (Aldh1). In this review, we discuss the role of Aldh1 enzymes in the generation of retinoic acid during adipogenesis, in the regulation of the transcriptional network of PPARγ in a fat-depot-specific manner, and the important contribution of this autocrine pathway in the development of visceral obesity. This article is part of a Special Issue entitled Retinoid and Lipid Metabolism.
Published by Elsevier B.V.
Figures

References
-
- Deitel M. Overweight and obesity worldwide now estimated to involve 1.7 billion people. Obes Surg. 2003;13:329–330. - PubMed
-
- Li C, Ford ES, McGuire LC, Mokdad AH. Increasing trends in waist circumference and abdominal obesity among US adults. Obesity (Silver Spring) 2007;15:216–224. - PubMed
-
- Zhang C, Rexrode KM, van Dam RM, Li TY, Hu FB. Abdominal obesity and the risk of all-cause, cardiovascular, and cancer mortality: sixteen years of follow-up in US women. Circulation. 2008;117:1658–1667. - PubMed
-
- Reis JP, Macera CA, Araneta MR, Lindsay SP, Marshall SJ, Wingard DL. Comparison of overall obesity and body fat distribution in predicting risk of mortality. Obesity (Silver Spring) 2009;17:1232–1239. - PubMed
-
- Chan JM, Rimm EB, Colditz GA, Stampfer MJ, Willett WC. Obesity, fat distribution, and weight gain as risk factors for clinical diabetes in men. Diabetes Care. 1994;17:961–969. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

