Prochelators triggered by hydrogen peroxide provide hexadentate iron coordination to impede oxidative stress
- PMID: 21708101
- PMCID: PMC3355344
- DOI: 10.1016/j.jinorgbio.2011.05.023
Prochelators triggered by hydrogen peroxide provide hexadentate iron coordination to impede oxidative stress
Abstract
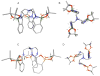
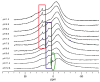
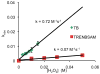
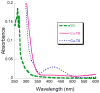
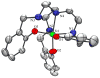
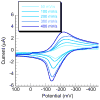
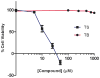
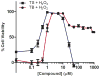
Prochelators are agents that have little affinity for metal ions until they undergo a chemical conversion. Three new aryl boronate prochelators are presented that are responsive to hydrogen peroxide to provide hexadentate ligands for chelating metal ions. TRENBSIM (tris[(2-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzylidene)-2-aminoethyl]amine), TRENBSAM (tris[(2-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzoyl)-2-aminoethyl]amine), and TB (tris[(2-boronic acid-benzyl)2-aminoethyl]amine) convert to TRENSIM (tris[(salicylideneamino)ethyl]amine), TRENSAM (tris[(2-hydroxybenzoyl)-2-aminoethyl]amine), and TS (tris[2-hydroxybenzyl)2-aminoethyl]amine), respectively. The prochelators were characterized by (11)B NMR, and the structures of TRENBSAM, TRENBSIM, and the Fe(III) complex of TS were determined by X-ray crystallography. Of the three prochelator/chelator pairs, TB/TS was identified as the most promising for biological applications, as they prevent iron and copper-induced hydroxyl radical generation in an in vitro assay. TB has negligible interactions with metal ions, whereas TS has apparent binding constants (log K') at pH 7.4 of 15.87 for Cu(II), 9.67 Zn(II) and 14.42 for Fe(III). Up to 1 mMTB was nontoxic to retinal pigment epithelial cells, whereas 10 μM TS induced cell death. TS protected cells against H(2)O(2)-induced death, but only within a 1-10 μM range. TB, on the other hand, had a much broader window of protection, suggesting that it may be a useful agent for preventing metal-promoted oxidative damage.
Copyright © 2011 Elsevier Inc. All rights reserved.
Figures
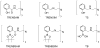
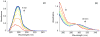

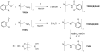
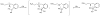
Similar articles
-
Modifying aroylhydrazone prochelators for hydrolytic stability and improved cytoprotection against oxidative stress.Bioorg Med Chem. 2018 Dec 1;26(22):5962-5972. doi: 10.1016/j.bmc.2018.11.004. Epub 2018 Nov 5. Bioorg Med Chem. 2018. PMID: 30429096 Free PMC article.
-
Prochelator BHAPI protects cells against paraquat-induced damage by ROS-triggered iron chelation.Metallomics. 2012 Aug;4(9):899-909. doi: 10.1039/c2mt20069d. Metallomics. 2012. PMID: 22700084 Free PMC article.
-
A pro-chelator triggered by hydrogen peroxide inhibits iron-promoted hydroxyl radical formation.J Am Chem Soc. 2006 Sep 27;128(38):12424-5. doi: 10.1021/ja064806w. J Am Chem Soc. 2006. PMID: 16984186
-
Prochelator strategies for site-selective activation of metal chelators.J Inorg Biochem. 2016 Sep;162:31-43. doi: 10.1016/j.jinorgbio.2016.05.012. Epub 2016 May 17. J Inorg Biochem. 2016. PMID: 27297691 Review.
-
Metals, toxicity and oxidative stress.Curr Med Chem. 2005;12(10):1161-208. doi: 10.2174/0929867053764635. Curr Med Chem. 2005. PMID: 15892631 Review.
Cited by
-
Stimulus-Responsive Prochelators for Manipulating Cellular Metals.Acc Chem Res. 2016 Nov 15;49(11):2468-2477. doi: 10.1021/acs.accounts.6b00380. Epub 2016 Oct 17. Acc Chem Res. 2016. PMID: 27749047 Free PMC article.
-
A boronate prochelator built on a triazole framework for peroxide-triggered tridentate metal binding.Inorganica Chim Acta. 2012 Dec 1;393:294-303. doi: 10.1016/j.ica.2012.06.011. Inorganica Chim Acta. 2012. PMID: 23439614 Free PMC article.
-
Cu2+ selective chelators relieve copper-induced oxidative stress in vivo.Chem Sci. 2018 Oct 2;9(41):7916-7930. doi: 10.1039/c8sc04041a. eCollection 2018 Nov 7. Chem Sci. 2018. PMID: 30450181 Free PMC article.
-
Boronate-Based Bioactive Compounds Activated by Peroxynitrite and Hydrogen Peroxide.Redox Biochem Chem. 2024 Dec;10:100040. doi: 10.1016/j.rbc.2024.100040. Epub 2024 Aug 14. Redox Biochem Chem. 2024. PMID: 39678628
-
Modifying aroylhydrazone prochelators for hydrolytic stability and improved cytoprotection against oxidative stress.Bioorg Med Chem. 2018 Dec 1;26(22):5962-5972. doi: 10.1016/j.bmc.2018.11.004. Epub 2018 Nov 5. Bioorg Med Chem. 2018. PMID: 30429096 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

