The mechanism of superoxide production by the antimycin-inhibited mitochondrial Q-cycle
- PMID: 21708945
- PMCID: PMC3173136
- DOI: 10.1074/jbc.M111.267898
The mechanism of superoxide production by the antimycin-inhibited mitochondrial Q-cycle
Abstract
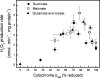
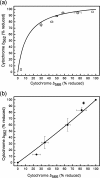
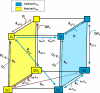
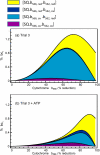
Superoxide production from antimycin-inhibited complex III in isolated mitochondria first increased to a maximum then decreased as substrate supply was modulated in three different ways. In each case, superoxide production had a similar bell-shaped relationship to the reduction state of cytochrome b(566), suggesting that superoxide production peaks at intermediate Q-reduction state because it comes from a semiquinone in the outer quinone-binding site in complex III (Q(o)). Imposition of a membrane potential changed the relationships between superoxide production and b(566) reduction and between b(562) and b(566) redox states, suggesting that b(562) reduction also affects semiquinone concentration and superoxide production. To assess whether this behavior was consistent with the Q-cycle mechanism of complex III, we generated a kinetic model of the antimycin-inhibited Q(o) site. Using published rate constants (determined without antimycin), with unknown rate constants allowed to vary, the model failed to fit the data. However, when we allowed the rate constant for quinol oxidation to decrease 1000-fold and the rate constant for semiquinone oxidation by b(566) to depend on the b(562) redox state, the model fit the energized and de-energized data well. In such fits, quinol oxidation was much slower than literature values and slowed further when b(566) was reduced, and reduction of b(562) stabilized the semiquinone when b(566) was oxidized. Thus, superoxide production at Q(o) depends on the reduction states of b(566) and b(562) and fits the Q-cycle only if particular rate constants are altered when b oxidation is prevented by antimycin. These mechanisms limit superoxide production and short circuiting of the Q-cycle when electron transfer slows.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

