Differential response of the human renal proximal tubular epithelial cell line HK-2 to Shiga toxin types 1 and 2
- PMID: 21708996
- PMCID: PMC3165488
- DOI: 10.1128/IAI.05139-11
Differential response of the human renal proximal tubular epithelial cell line HK-2 to Shiga toxin types 1 and 2
Abstract
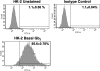
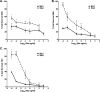
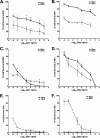
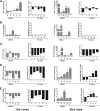
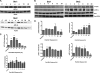
Shiga toxins (Stxs) are expressed by the enteric pathogens Shigella dysenteriae serotype 1 and certain serotypes of Escherichia coli. Stx-producing bacteria cause bloody diarrhea with the potential to progress to acute renal failure. Stxs are potent protein synthesis inhibitors and are the primary virulence factors responsible for renal damage that may follow diarrheal disease. We explored the use of the immortalized human proximal tubule epithelial cell line HK-2 as an in vitro model of Stx-induced renal damage. We showed that these cells express abundant membrane Gb(3) and are differentially susceptible to the cytotoxic action of Stxs, being more sensitive to Shiga toxin type 1 (Stx1) than to Stx2. At early time points (24 h), HK-2 cells were significantly more sensitive to Stxs than Vero cells; however, by 72 h, Vero cell monolayers were completely destroyed while some HK-2 cells survived toxin challenge, suggesting that a subpopulation of HK-2 cells are relatively toxin resistant. Fluorescently labeled Stx1 B subunits localized to both lysosomal and endoplasmic reticulum (ER) compartments in HK-2 cells, suggesting that differences in intracellular trafficking may play a role in susceptibility to Stx-mediated cytotoxicity. Although proinflammatory cytokines were not upregulated by toxin challenge, Stx2 selectively induced the expression of two chemokines, macrophage inflammatory protein-1α (MIP-1α) and MIP-1β. Stx1 and Stx2 differentially activated components of the ER stress response in HK-2 cells. Finally, we demonstrated significant poly(ADP-ribose) polymerase (PARP) cleavage after exposure to Stx1 or Stx2. However, procaspase 3 cleavage was undetectable, suggesting that HK-2 cells may undergo apoptosis in response to Stxs in a caspase 3-independent manner.
Figures



Similar articles
-
Shiga Toxins Induce Apoptosis and ER Stress in Human Retinal Pigment Epithelial Cells.Toxins (Basel). 2017 Oct 13;9(10):319. doi: 10.3390/toxins9100319. Toxins (Basel). 2017. PMID: 29027919 Free PMC article.
-
Cytoprotective effect of curcumin in human proximal tubule epithelial cells exposed to shiga toxin.Biochem Biophys Res Commun. 2001 Apr 27;283(1):36-41. doi: 10.1006/bbrc.2001.4749. Biochem Biophys Res Commun. 2001. PMID: 11322764
-
Comparative evaluation of apoptosis induced by Shiga toxin 1 and/or lipopolysaccharides in human monocytic and macrophage-like cells.Microb Pathog. 2005 Feb-Mar;38(2-3):63-76. doi: 10.1016/j.micpath.2004.12.003. Microb Pathog. 2005. PMID: 15748808
-
Shiga toxins and apoptosis.FEMS Microbiol Lett. 2003 Nov 21;228(2):159-66. doi: 10.1016/S0378-1097(03)00761-4. FEMS Microbiol Lett. 2003. PMID: 14638419 Review.
-
Shiga Toxin (Stx) Classification, Structure, and Function.Microbiol Spectr. 2014 Aug;2(4):EHEC-0024-2013. doi: 10.1128/microbiolspec.EHEC-0024-2013. Microbiol Spectr. 2014. PMID: 25530917 Free PMC article. Review.
Cited by
-
Shiga Toxins as Antitumor Tools.Toxins (Basel). 2021 Sep 28;13(10):690. doi: 10.3390/toxins13100690. Toxins (Basel). 2021. PMID: 34678982 Free PMC article. Review.
-
Shiga Toxin Therapeutics: Beyond Neutralization.Toxins (Basel). 2017 Sep 19;9(9):291. doi: 10.3390/toxins9090291. Toxins (Basel). 2017. PMID: 28925976 Free PMC article. Review.
-
Recent Updates on Outbreaks of Shiga Toxin-Producing Escherichia coli and Its Potential Reservoirs.Front Cell Infect Microbiol. 2020 Jun 4;10:273. doi: 10.3389/fcimb.2020.00273. eCollection 2020. Front Cell Infect Microbiol. 2020. PMID: 32582571 Free PMC article. Review.
-
Structural Insights into Escherichia coli Shiga Toxin (Stx) Glycosphingolipid Receptors of Porcine Renal Epithelial Cells and Inhibition of Stx-Mediated Cellular Injury Using Neoglycolipid-Spiked Glycovesicles.Microorganisms. 2019 Nov 19;7(11):582. doi: 10.3390/microorganisms7110582. Microorganisms. 2019. PMID: 31752441 Free PMC article.
-
Shiga Toxins Activate the NLRP3 Inflammasome Pathway To Promote Both Production of the Proinflammatory Cytokine Interleukin-1β and Apoptotic Cell Death.Infect Immun. 2015 Oct 26;84(1):172-86. doi: 10.1128/IAI.01095-15. Print 2016 Jan. Infect Immun. 2015. PMID: 26502906 Free PMC article.
References
-
- Bauwens A., et al. 2011. Differential cytotoxic actions of Shiga toxin 1 and Shiga toxin 2 on microvascular and macrovascular endothelial cells. Thromb. Haemost. 105:515–528 - PubMed
-
- Bernales S., Papa F. R., Walter P. 2006. Intracellular signaling by the unfolded protein response. Annu. Rev. Cell Dev. Biol. 22:487–508 - PubMed
-
- Breckenridge D. G., Germain M., Mathai J. P., Nguyen M., Shore G. C. 2003. Regulation of apoptosis by endoplasmic reticulum pathways. Oncogene 22:8608–8618 - PubMed
-
- Brigotti M., et al. 2008. Interactions between Shiga toxins and human polymorphonuclear leukocytes. J. Leukoc. Biol. 84:1019–1027 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

