A small amount of fat does not affect piperaquine exposure in patients with malaria
- PMID: 21709087
- PMCID: PMC3165307
- DOI: 10.1128/AAC.00279-11
A small amount of fat does not affect piperaquine exposure in patients with malaria
Abstract
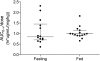
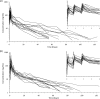
Dihydroartemisinin-piperaquine is a new, highly effective, and well-tolerated combination treatment for uncomplicated falciparum malaria. The lipophilic characteristic of piperaquine suggests that administration together with fat will increase the oral bioavailability of the drug, and this has been reported for healthy volunteers. This pharmacokinetic study monitored 30 adult patients with uncomplicated falciparum malaria for 4.5 months to evaluate the effects of the concomitant intake of fat on the total piperaquine exposure. The fixed-drug combination of dihydroartemisinin-piperaquine was given with water to fasting patients (n = 15) or was coadministered with 200 ml milk containing 6.4 g fat (n = 15). The drug combination was generally well tolerated, and there were no severe adverse effects reported for either group during the study. Total piperaquine exposure (area under the concentration-time curve from zero to infinity [AUC(0-∞)]; results are given as medians [ranges]) were not statistically different between fed (29.5 h · μg/ml [20.6 to 58.7 h · μg/ml]) and fasting (23.9 h · μg/ml [11.9 to 72.9 h · μg/ml]) patients, but the interindividual variation was reduced in the fed group. Overall, none of the pharmacokinetic parameters differed statistically between the groups. Total piperaquine exposure correlated well with the day 7 concentrations in the fasted group, but the fed group showed a poor correlation. In conclusion, the coadministration of 6.4 g fat did not have any significant effect on piperaquine pharmacokinetics in the treatment of uncomplicated malaria.
Figures

References
-
- Ahmed T., et al. 2008. Safety, tolerability, and single- and multiple-dose pharmacokinetics of piperaquine phosphate in healthy subjects. J. Clin. Pharmacol. 48:166–175 - PubMed
-
- Ashley E. A., et al. 2005. A randomized, controlled study of a simple, once-daily regimen of dihydroartemisinin-piperaquine for the treatment of uncomplicated, multidrug-resistant falciparum malaria. Clin. Infect. Dis. 41:425–432 - PubMed
-
- Ashley E. A., et al. 2007. How much fat is necessary to optimize lumefantrine oral bioavailability? Trop. Med. Int. Health 12:195–200 - PubMed
-
- Brockman A., et al. 1999. Application of genetic markers to the identification of recrudescent Plasmodium falciparum infections on the northwestern border of Thailand. Am. J. Trop. Med. Hyg. 60:14–21 - PubMed
-
- Chen L., Qu F. Y., Zhou Y. C. 1982. Field observations on the antimalarial piperaquine. Chin. Med. J. 95:281–286 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

