Lack of macrophage migration inhibitory factor in mice does not affect hallmarks of the inflammatory/immune response during the first week after stroke
- PMID: 21714902
- PMCID: PMC3152909
- DOI: 10.1186/1742-2094-8-75
Lack of macrophage migration inhibitory factor in mice does not affect hallmarks of the inflammatory/immune response during the first week after stroke
Abstract
Background: Macrophage migration inhibitory factor (MIF) has been proposed to play a detrimental role in stroke. We recently showed that MIF promotes neuronal death and aggravates neurological deficits during the first week after experimental stroke, in mice. Since MIF regulates tissue inflammation, we studied the putative role of MIF in post-stroke inflammation.
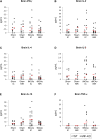
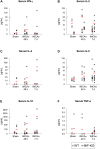
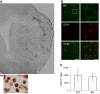
Methods: We subjected C57BL/6 mice, Mif-/- (MIF-KO) or Mif+/+ (WT), to a transient occlusion of the right middle cerebral artery (tMCAo) or sham-surgery. We studied MIF expression, GFAP expression and the number of CD74-positive cells in the ischemic brain hemisphere 7 days after tMCAo using primarily immunohistochemistry. We determined IFN-γ, IL-2, IL-4, IL-5, IL-10, IL-12, KC/CXCL-1 and TNF-α protein levels in the brain (48 h after surgery) and serum (48 h and 7 days after surgery) by a multiplex immunoassay.
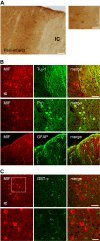
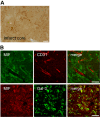
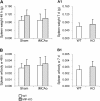
Results: We observed that MIF accumulates in neurons and astrocytes of the peri-infarct region, as well as in microglia/macrophages of the infarct core up to 7 days after stroke. Among the inflammatory mediators analyzed, we found a significant increase in cerebral IL-12 and KC levels after tMCAo, in comparison to sham-surgery. Importantly, the deletion of Mif did not significantly affect the levels of the cytokines evaluated, in the brain or serum. Moreover, the spleen weight 48 h and 7 days subsequent to tMCAo was similar in WT and MIF-KO mice. Finally, the extent of GFAP immunoreactivity and the number of MIF receptor (CD74)-positive cells within the ischemic brain hemisphere did not differ significantly between WT and MIF-KO mice subjected to tMCAo.
Conclusions: We conclude that MIF does not affect major components of the inflammatory/immune response during the first week after experimental stroke. Based on present and previous evidence, we propose that the deleterious MIF-mediated effects in stroke depend primarily on an intraneuronal and/or interneuronal action.
Figures


References
-
- Liu T, McDonnell PC, Young PR, White RF, Siren AL, Hallenbeck JM, Barone FC, Feurestein GZ. Interleukin-1 beta mRNA expression in ischemic rat cortex. Stroke. 1993;24:1746–50. - PubMed
-
- Yamasaki Y, Matsuura N, Shozuhara H, Onodera H, Itoyama Y, Kogure K. Interleukin-1 as a pathogenetic mediator of ischemic brain damage in rats. Stroke. 1995;26:676–80. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

