Importance of the CCR5-CCL5 axis for mucosal Trypanosoma cruzi protection and B cell activation
- PMID: 21715689
- PMCID: PMC3150841
- DOI: 10.4049/jimmunol.1100033
Importance of the CCR5-CCL5 axis for mucosal Trypanosoma cruzi protection and B cell activation
Abstract
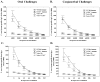
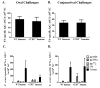
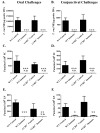
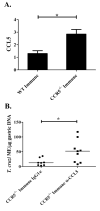
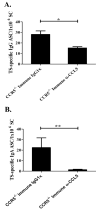
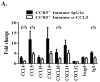
Trypanosoma cruzi is an intracellular parasite and the causative agent of Chagas disease. Previous work has shown that the chemokine receptor CCR5 plays a role in systemic T. cruzi protection. We evaluated the importance of CCR5 and CCL5 for mucosal protection against natural oral and conjunctival T. cruzi challenges. T. cruzi-immune CCR5(-/-) and wild-type C57BL/6 mice were generated by repeated infectious challenges with T. cruzi. CCR5(-/-) and wild-type mice developed equivalent levels of cellular, humoral, and protective mucosal responses. However, CCR5(-/-)-immune mice produced increased levels of CCL5 in protected gastric tissues, suggesting compensatory signaling through additional receptors. Neutralization of CCL5 in CCR5(-/-)-immune mice resulted in decreased mucosal inflammatory responses, reduced T. cruzi-specific Ab-secreting cells, and significantly less mucosal T. cruzi protection, confirming an important role for CCL5 in optimal immune control of T. cruzi replication at the point of initial mucosal invasion. To investigate further the mechanism responsible for mucosal protection mediated by CCL5-CCR5 signaling, we evaluated the effects of CCL5 on B cells. CCL5 enhanced proliferation and IgM secretion in highly purified B cells triggered by suboptimal doses of LPS. In addition, neutralization of endogenous CCL5 inhibited B cell proliferation and IgM secretion during stimulation of highly purified B cells, indicating that B cell production of CCL5 has important autocrine effects. These findings demonstrate direct effects of CCL5 on B cells, with significant implications for the development of mucosal adjuvants, and further suggest that CCL5 may be important as a general B cell coactivator.
Figures

Similar articles
-
Deficiency of antigen-specific B cells results in decreased Trypanosoma cruzi systemic but not mucosal immunity due to CD8 T cell exhaustion.J Immunol. 2015 Feb 15;194(4):1806-18. doi: 10.4049/jimmunol.1303163. Epub 2015 Jan 16. J Immunol. 2015. PMID: 25595788 Free PMC article.
-
Th17 Cells Provide Mucosal Protection against Gastric Trypanosoma cruzi Infection.Infect Immun. 2021 Jun 16;89(7):e0073820. doi: 10.1128/IAI.00738-20. Epub 2021 Jun 16. Infect Immun. 2021. PMID: 33941576 Free PMC article.
-
The CC chemokine receptor 5 is important in control of parasite replication and acute cardiac inflammation following infection with Trypanosoma cruzi.Infect Immun. 2006 Jan;74(1):135-43. doi: 10.1128/IAI.74.1.135-143.2006. Infect Immun. 2006. PMID: 16368966 Free PMC article.
-
Genetic Polymorphism at CCL5 Is Associated With Protection in Chagas' Heart Disease: Antagonistic Participation of CCR1+ and CCR5+ Cells in Chronic Chagasic Cardiomyopathy.Front Immunol. 2018 Apr 11;9:615. doi: 10.3389/fimmu.2018.00615. eCollection 2018. Front Immunol. 2018. PMID: 29696014 Free PMC article.
-
Chemokine receptor 5 signaling in oral diseases and degenerative temporomandibular joint disease.J Oral Biosci. 2025 Jun;67(2):100666. doi: 10.1016/j.job.2025.100666. Epub 2025 Apr 24. J Oral Biosci. 2025. PMID: 40287135 Review.
Cited by
-
New Data on Human Macrophages Polarization by Hymenolepis diminuta Tapeworm-An In Vitro Study.Front Immunol. 2017 Feb 20;8:148. doi: 10.3389/fimmu.2017.00148. eCollection 2017. Front Immunol. 2017. PMID: 28265273 Free PMC article.
-
Deficiency of antigen-specific B cells results in decreased Trypanosoma cruzi systemic but not mucosal immunity due to CD8 T cell exhaustion.J Immunol. 2015 Feb 15;194(4):1806-18. doi: 10.4049/jimmunol.1303163. Epub 2015 Jan 16. J Immunol. 2015. PMID: 25595788 Free PMC article.
-
Trypanosoma cruzi Entrance through Systemic or Mucosal Infection Sites Differentially Modulates Regional Immune Response Following Acute Infection in Mice.Front Immunol. 2013 Jul 26;4:216. doi: 10.3389/fimmu.2013.00216. eCollection 2013. Front Immunol. 2013. PMID: 23898334 Free PMC article.
-
The protective and pathogenic roles of CXCL17 in human health and disease: Potential in respiratory medicine.Cytokine Growth Factor Rev. 2020 Jun;53:53-62. doi: 10.1016/j.cytogfr.2020.04.004. Epub 2020 Apr 23. Cytokine Growth Factor Rev. 2020. PMID: 32345516 Free PMC article. Review.
-
Host Genetics Background Influence in the Intragastric Trypanosoma cruzi Infection.Front Immunol. 2020 Nov 24;11:566476. doi: 10.3389/fimmu.2020.566476. eCollection 2020. Front Immunol. 2020. PMID: 33329529 Free PMC article.
References
-
- Centers for Disease, C., and Prevention. Chagas Disease; Epidemiology & Risk Factors. 2007 http://www.cdc.gov/chagas/epi.html.
-
- Bern C, Montgomery SP. An estimate of the burden of Chagas disease in the United States. Clinical Infectious Diseases. 2009;49:e52–e54. - PubMed
-
- Allen SJ, Crown SE, Handel TM. Chemokine: receptor structure, interactions, and antagonism. Annual Review of Immunology. 2007;25:787–820. - PubMed
-
- Honczarenko M, Le Y, Glodek AM, Majka M, Campbell JJ, Ratajczak MZ, Silberstein LE. CCR5-binding chemokines modulate CXCL12 (SDF-1)-induced responses of progenitor B cells in human bone marrow through heterologous desensitization of the CXCR4 chemokine receptor. Blood. 2002;100:2321–2329. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

