Of mice and not humans: how reliable are animal models for evaluation of herpes CD8(+)-T cell-epitopes-based immunotherapeutic vaccine candidates?
- PMID: 21718746
- PMCID: PMC3159167
- DOI: 10.1016/j.vaccine.2011.06.083
Of mice and not humans: how reliable are animal models for evaluation of herpes CD8(+)-T cell-epitopes-based immunotherapeutic vaccine candidates?
Abstract
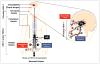
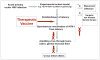
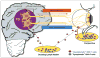
Herpes simplex virus type 1 and type 2 (HSV-1 and HSV-2)-specific CD8(+) T cells that reside in sensory ganglia, appear to control recurrent herpetic disease by aborting or reducing spontaneous and sporadic reactivations of latent virus. A reliable animal model is the ultimate key factor to test the efficacy of therapeutic vaccines that boost the level and the quality of sensory ganglia-resident CD8(+) T cells against spontaneous herpes reactivation from sensory neurons, yet its relevance has been often overlooked. Herpes vaccinologists are hesitant about using mouse as a model in pre-clinical development of therapeutic vaccines because they do not adequately mimic spontaneous viral shedding or recurrent symptomatic diseases, as occurs in human. Alternatives to mouse models are rabbits and guinea pigs in which reactivation arise spontaneously with clinical herpetic features relevant to human disease. However, while rabbits and guinea pigs develop spontaneous HSV reactivation and recurrent ocular and genital disease none of them can mount CD8(+) T cell responses specific to Human Leukocyte Antigen- (HLA-)restricted epitopes. In this review, we discuss the advantages and limitations of these animal models and describe a novel "humanized" HLA transgenic rabbit, which shows spontaneous HSV-1 reactivation, recurrent ocular disease and mounts CD8(+) T cell responses to HLA-restricted epitopes. Adequate investments are needed to develop reliable preclinical animal models, such as HLA class I and class II double transgenic rabbits and guinea pigs to balance the ethical and financial concerns associated with the rising number of unsuccessful clinical trials for therapeutic vaccine formulations tested in unreliable mouse models.
Copyright © 2011 Elsevier Ltd. All rights reserved.
Figures

References
-
- Morrison LA. Vaccines against genital herpes: progress and limitations. Drugs. 2002;62(8):1119–29. - PubMed
-
- Wilson KM, Triantafilou K, Morrison IE, Cherry RJ, Fernandez N. Single particle imaging of cell-surface HLA-DR tetramers. Biochem Soc Trans. 1997 May;25(2):360S. - PubMed
-
- Rubbo PA, Tuaillon E, Nagot N, Chentoufi AA, Bollore K, REYNES J, et al. HIV-1 infection impairs HSV-specific CD4+ and CD8+ T cell response by reducing Th1 cytokines and CCR5 ligand secretion. AIDS. 2011 In press. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

