IL-25 induces M2 macrophages and reduces renal injury in proteinuric kidney disease
- PMID: 21719780
- PMCID: PMC3137571
- DOI: 10.1681/ASN.2010070693
IL-25 induces M2 macrophages and reduces renal injury in proteinuric kidney disease
Abstract
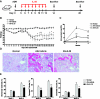
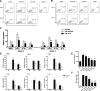
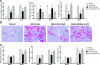
The kidney contains receptors for the cytokine IL-25, but the effects of IL-25 in CKD are unknown. Here, we induced adriamycin nephropathy in both BALB/c mice and severe combined immunodeficient (SCID) mice, and we injected IL-25 for 7 consecutive days starting at day 5 after adriamycin administration. BALB/c mice treated with IL-25 had less glomerulosclerosis, tubular atrophy, interstitial expansion, and proteinuria than control mice at day 28. IL-25 increased the levels of IL-4 and IL-13 in serum, kidney, renal draining lymph nodes, and CD4+ lymphocytes. IL-25 also directly suppressed effector macrophages in vitro and in vivo and induced alternatively activated (M2) macrophages in vivo. However, in SCID mice and in BALB/c mice treated with IL-4/13-neutralizing antibody, IL-25 failed to protect against renal injury and did not induce M2. In conclusion, IL-25 protects against renal injury in adriamycin nephropathy in mice by, at least in part, inducing Th2 immune responses.
Copyright © 2011 by the American Society of Nephrology
Figures
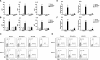



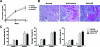
References
-
- Moseley TA, Haudenschild DR, Rose L, Reddi AH: Interleukin-17 family and IL-17 receptors. Cytokine Growth Factor Rev 14: 155–174, 2003 - PubMed
-
- Ikeda K, Nakajima H, Suzuki K, Kagami S, Hirose K, Suto A, Saito Y, Iwamoto I: Mast cells produce interleukin-25 upon Fc epsilon RI-mediated activation. Blood 101: 3594–3596, 2003 - PubMed
-
- Fort MM, Cheung J, Yen D, Li J, Zurawski SM, Lo S, Menon S, Clifford T, Hunte B, Lesley R, Muchamuel T, Hurst SD, Zurawski G, Leach MW, Gorman DM, Rennick DM: IL-25 induces IL-4, IL-5, and IL-13 and Th2-associated pathologies in vivo. Immunity 15: 985–995, 2001 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

