miR-335 and miR-34a Promote renal senescence by suppressing mitochondrial antioxidative enzymes
- PMID: 21719785
- PMCID: PMC3137573
- DOI: 10.1681/ASN.2010040367
miR-335 and miR-34a Promote renal senescence by suppressing mitochondrial antioxidative enzymes
Abstract
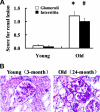
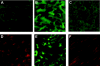
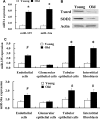
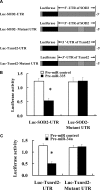
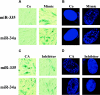
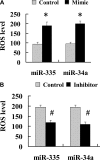
The molecular basis for aging of the kidney is not well understood. MicroRNAs (miRNAs) contribute to processes such as development, differentiation, and apoptosis, but their contribution to the aging process is unknown. Here, we analyzed the miRNA expression profile of young (3-month) and old (24-month) rat kidneys and identified the biologic pathways and genes regulated by differentially expressed miRNAs. We observed upregulation of 18 miRNAs with aging, mainly regulating the genes associated with energy metabolism, cell proliferation, antioxidative defense, and extracellular matrix degradation; in contrast, we observed downregulation of 7 miRNAs with aging, principally targeting the genes associated with the immune inflammatory response and cell-cycle arrest. Bioinformatics analysis suggested that superoxide dismutase 2 (SOD2) and thioredoxin reductase 2 (Txnrd2), located in the mitochondria, are potential targets of miR-335 and miR-34a, respectively. Aging mesangial cells exhibited significant upregulation of miR-335 and miR-34a and marked downregulation of SOD2 and Txnrd2. miR-335 and miR-34a inhibited expression of SOD2 and Txnrd2 by binding to the 3'-untranslated regions of each gene, respectively. Overexpression of miR-335 and miR-34a induced premature senescence of young mesangial cells via suppression of SOD2 and Txnrd2 with a concomitant increase in reactive oxygen species (ROS). Conversely, antisense miR-335 and miR-34a inhibited senescence of old mesangial cells via upregulation of SOD2 and Txnrd2 with a concomitant decrease in ROS. In conclusion, these results suggest that miRNAs may contribute to renal aging by inhibiting intracellular pathways such as those involving the mitochondrial antioxidative enzymes SOD2 and Txnrd2.
Copyright © 2011 by the American Society of Nephrology
Figures


References
-
- Melk A, Mansfield ES, Hsieh SC, Hernandez-Boussard T, Grimm P, Rayner DC, Halloran PF, Sarwal MM: Transcriptional analysis of the molecular basis of human kidney aging using cDNA microarray profiling. Kidney Int 68: 2667–2679, 2005 - PubMed
-
- Calin GA, Croce MA: MicroRNA-cancer connection: The beginning of a new tale. Cancer Res 66: 7390–7394, 2006 - PubMed
-
- Yang B, Lin H, Xiao J, Lu Y, Luo X, Li B, Zhang Y, Xu C, Bai Y, Wang H, Chen G, Wang Z: The muscle-specific microRNA miR-1 regulates cardiac arrhythmogenic potential by targeting GJA1 and KCNJ2. Nature Med 13: 486–491, 2007 - PubMed
-
- Lewis BP, Burge CB, Bartel DP: Conserved seed pairing, often flanked by adenosines, indicates that thousands of human genes are microRNA targets. Cell 120: 15–20, 2005 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

