How ubiquitin functions with ESCRTs
- PMID: 21722280
- PMCID: PMC3171646
- DOI: 10.1111/j.1600-0854.2011.01242.x
How ubiquitin functions with ESCRTs
Abstract
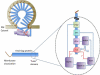
The endosomal-sorting complex required for transport (ESCRT) apparatus has multiple ubiquitin (Ub)-binding domains and participates in a wide variety of cellular processes. Many of these ESCRT-dependent processes are keenly regulated by Ub, which serves as a lysosomal-sorting signal for membrane proteins targeted into multivesicular bodies (MVBs) and which may serve as a mediator of viral budding from the cell surface. Hints that both ESCRTs and Ub work together in the processes such as cytokinesis, transcription and autophagy are beginning to emerge. Here, we explore the relationship between ESCRTs and Ub in MVB sorting and viral budding.
© 2011 John Wiley & Sons A/S.
Figures

References
-
- Odorizzi G, Babst M, Emr SD. Fab1p PtdIns(3)P 5-kinase function essential for protein sorting in the multivesicular body. Cell. 1998;95(6):847–858. - PubMed
-
- Clague MJ, Urbe S. Ubiquitin: same molecule, different degradation pathways. Cell. 2010;143(5):682–685. - PubMed
-
- Raiborg C, Stenmark H. The ESCRT machinery in endosomal sorting of ubiquitylated membrane proteins. Nature. 2009;458(7237):445–452. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

