T4 RNA ligase 2 truncated active site mutants: improved tools for RNA analysis
- PMID: 21722378
- PMCID: PMC3149579
- DOI: 10.1186/1472-6750-11-72
T4 RNA ligase 2 truncated active site mutants: improved tools for RNA analysis
Abstract
Background: T4 RNA ligases 1 and 2 are useful tools for RNA analysis. Their use upstream of RNA analyses such as high-throughput RNA sequencing and microarrays has recently increased their importance. The truncated form of T4 RNA ligase 2, comprising amino acids 1-249 (T4 Rnl2tr), is an attractive tool for attachment of adapters or labels to RNA 3'-ends. Compared to T4 RNA ligase 1, T4 Rnl2tr has a decreased ability to ligate 5'-PO4 ends in single-stranded RNA ligations, and compared to the full-length T4 Rnl2, the T4 Rnl2tr has an increased activity for joining 5'-adenylated adapters to RNA 3'-ends. The combination of these properties allows adapter attachment to RNA 3'-ends with reduced circularization and concatemerization of substrate RNA.
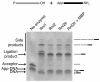
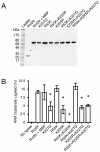
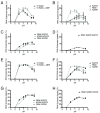
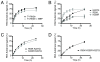
Results: With the aim of further reducing unwanted side ligation products, we substituted active site residues, known to be important for adenylyltransferase steps of the ligation reaction, in the context of T4 Rnl2tr. We characterized the variant ligases for the formation of unwanted ligation side products and for activity in the strand-joining reaction.
Conclusions: Our data demonstrate that lysine 227 is a key residue facilitating adenylyl transfer from adenylated ligation donor substrates to the ligase. This reversal of the second step of the ligation reaction correlates with the formation of unwanted ligation products. Thus, T4 Rn2tr mutants containing the K227Q mutation are useful for reducing undesired ligation products. We furthermore report optimal conditions for the use of these improved T4 Rnl2tr variants.
Figures




References
-
- Ho CK, Wang LK, Lima CD, Shuman S. Structure and mechanism of RNA ligase. Structure. 2004;12:327–339. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

