Efficient Expression of Bioactive Human Leptin in Escherichia coli in Soluble Fusion Form
- PMID: 21731206
- PMCID: PMC3001832
- DOI: 10.1007/s12291-010-0066-2
Efficient Expression of Bioactive Human Leptin in Escherichia coli in Soluble Fusion Form
Abstract
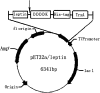
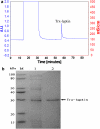
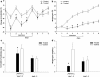
Leptin, a 16 kDa nonglycosylated hormone, is produced by mature adipocytes and functions primarily in the hypothalamus to reduce food intake and body weight. To explore a new approach for high-level expression of human Leptin in Escherichia coli, the human Leptin gene, synthesized according to the published sequence, was cloned into the vector pET32a to construct a fusion expression plasmid: Trx-Leptin/pET32a. Our data showed that more than 40% of the fusion protein Trx-Leptin was expressed in soluble form. After purified by Ni-IDA affinity chromatography, cleaved by enterokinase and applied Ni-IDA affinity chromatography again, purified Leptin with homogeneity over 96% was achieved. The bio-functional experiments of purified Leptin showed a significant reduction in food intake and body weight of female mice treated with Leptin by comparing with control mice, and it indicated that the purified Leptin has full biological activity. In addition, our expression system was a very low-cost and efficient prokaryotic expression system. So taken together, our results demonstrated that our expression system of bio-active Leptin provided a new method for producing Leptin in big scale and would be widely applied in commercial Leptin producing industries.
Keywords: Affinity purification; Enterokinase; Escherichia coli; Fusion expression; Human leptin; Trx.
Figures


Similar articles
-
Recombinant expression of biologically active rat leptin in Escherichia coli.Protein Expr Purif. 2001 Jun;22(1):60-9. doi: 10.1006/prep.2001.1412. Protein Expr Purif. 2001. PMID: 11388800
-
Expression of recombinant hybrid peptide hinnavin II/alpha-melanocyte-stimulating hormone in Escherichia coli: purification and characterization.J Microbiol. 2010 Feb;48(1):24-9. doi: 10.1007/s12275-009-0317-1. Epub 2010 Mar 11. J Microbiol. 2010. PMID: 20221725
-
Expression in Escherichia coli and purification of bioactive antibacterial peptide ABP-CM4 from the Chinese silk worm, Bombyx mori.Biotechnol Lett. 2007 Jul;29(7):1031-6. doi: 10.1007/s10529-007-9351-4. Epub 2007 Mar 21. Biotechnol Lett. 2007. PMID: 17375264
-
High-level expression, purification and characterization of active human C1q and tumour necrosis factor-related protein-1 in Escherichia coli.Lett Appl Microbiol. 2014 Sep;59(3):334-41. doi: 10.1111/lam.12280. Epub 2014 Jun 14. Lett Appl Microbiol. 2014. PMID: 24814641
-
High level expression, purification and characterization of active fusion human C1q and tumor necrosis factor related protein 2 (hCTRP2) in Escherichia coli.Protein Expr Purif. 2011 Sep;79(1):1-6. doi: 10.1016/j.pep.2011.03.013. Epub 2011 Mar 29. Protein Expr Purif. 2011. PMID: 21453774 Review.
Cited by
-
Engineered E. coli Nissle 1917 for delivery of bioactive IL-2 for cancer immunotherapy.Sci Rep. 2023 Aug 2;13(1):12506. doi: 10.1038/s41598-023-39365-2. Sci Rep. 2023. PMID: 37532747 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources
