Characterization of DISC formation and TNFR1 translocation to mitochondria in TNF-α-treated hepatocytes
- PMID: 21741934
- PMCID: PMC3157172
- DOI: 10.1016/j.ajpath.2011.05.046
Characterization of DISC formation and TNFR1 translocation to mitochondria in TNF-α-treated hepatocytes
Abstract
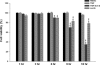
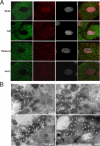
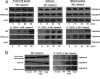
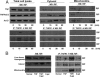
Tumor necrosis factor receptor 1 (TNFR1) activation in hepatocytes can trigger apoptotic or inflammatory signaling. The factors that determine which signaling pathway dominates are not clear and are thought to relate to the efficiency of death-inducing signaling complex (DISC) formation. However, the steps involved in DISC formation in hepatocytes are poorly understood. In characterizing DISC formation within cultured hepatocytes, we demonstrated that TNF-α exposure leads to the rapid formation of a DISC involving TNF-α, the TNFR-associated death domain adaptor molecule (TRADD), the Fas-associated death domain adaptor molecule (FADD), caspase-8, TNFR-associated factor 2 (TRAF2), and receptor-interacting protein (RIP). The inclusion of the sensitizing agent actinomycin D both accelerated and amplified the appearance of the DISC. Notably, TNFR1 along with some DISC components also appeared within mitochondria within 30 minutes. Whereas TNFR1 consistently co-localized with the TRADD, FADD, the caspase-8, and TRAF2 in the cytosolic fraction, TNFR1 in the mitochondria was associated only with caspase-8 after TNF-α exposure. Similar observations were made in vivo using TNF-α with D-galactosamine. Actinomycin D alone also enhanced the appearance of DISC components in both cytosol and the mitochondria. Thus the DISC that includes TNFR1 forms in the cytosol of hepatocytes under both survival and pro-apoptotic conditions. The observations also suggest that TNF-α-mediated signaling includes the translocation of TNFR1 to mitochondria.
Copyright © 2011 American Society for Investigative Pathology. Published by Elsevier Inc. All rights reserved.
Figures




References
-
- Rudiger H.A., Clavien P.A. Tumor necrosis factor alpha, but not Fas, mediates hepatocellular apoptosis in the murine ischemic liver. Gastroenterology. 2002;122:202–210. - PubMed
-
- Simeonova P.P., Gallucci R.M., Hulderman T., Wilson R., Kommineni C., Rao M., Luster M.I. The role of tumor necrosis factor-alpha in liver toxicity, inflammation, and fibrosis induced by carbon tetrachloride. Toxicol Appl Pharmacol. 2001;177:112–120. - PubMed
-
- Yin M., Wheeler M.D., Kono H., Bradford B.U., Gallucci R.M., Luster M.I., Thurman R.G. Essential role of tumor necrosis factor alpha in alcohol-induced liver injury in mice. Gastroenterology. 1999;117:942–952. - PubMed
-
- Leist M., Gantner F., Jilg S., Wendel A. Activation of the 55 kDa TNF-α receptor is necessary and sufficient for TNF-α-induced liver failure, hepatocyte apoptosis, and nitrite release. J Immunol. 1995;154:1307–1316. - PubMed
-
- Garcia-Ruiz C., Fernandez-Checa J.C. Redox regulation of hepatocyte apoptosis. J Gastroenterol Hepatol. 2007;22(Suppl 1):S38–S42. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

