Principles of electrospray ionization
- PMID: 21742801
- PMCID: PMC3134074
- DOI: 10.1074/mcp.M111.009407
Principles of electrospray ionization
Abstract
Electrospray ionization is today the most widely used ionization technique in chemical and biochemical analysis. Interfaced with a mass spectrometer it allows the investigation of the molecular composition of liquid samples. With electrospray a large variety of chemical substances can be ionized. There is no limitation in mass which thus enables even the investigation of large noncovalent protein complexes. Its high ionization efficiency profoundly changed biomolecular sciences because proteins can be identified and quantified on trace amounts in a high throughput fashion. This review article focuses mainly on the exploration of the underlying ionization mechanism. Some ionization characteristics are discussed that are related to this mechanism. Typical spectra of peptides, proteins, and noncovalent complexes are shown and the quantitative character of spectra is highlighted. Finally the possibilities and limitations in measuring the association constant of bivalent noncovalent complexes are described.
Figures
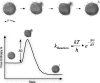

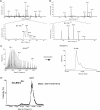
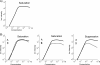

References
-
- Karas M., Hillenkamp F. (1988) Laser desorption ionization of proteins with molecular masses exceeding 10,000 daltons. Anal. Chem. 60, 2299–2301 - PubMed
-
- Meng C. K., Mann M., Fenn J. (1988) Of protons or proteins. Zeitschrift für Physik D Atoms, Molecules and Clusters 10, 361–368
-
- Fenn J. B., Mann M., Meng C. K., Wong S. F., Whitehouse C. M. (1989) Electrospray ionization for mass spectrometry of large biomolecules. Science 246, 64–71 - PubMed
-
- Taylor G. (1964) Disintegration of water drops in an electric field. Proc. R. Soc. Lon. A 280, 383–397
-
- Dole M., Mack L. L., Hines R. L., Mobley R. C., Ferguson L. D., Alice M. B. (1968) Molecular beams of macroions. J. Chem. Phys. 49, 2240–2249
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

