Estimating the pKa values of basic and acidic side chains in ion channels using electrophysiological recordings: a robust approach to an elusive problem
- PMID: 21744391
- PMCID: PMC7036269
- DOI: 10.1002/prot.23087
Estimating the pKa values of basic and acidic side chains in ion channels using electrophysiological recordings: a robust approach to an elusive problem
Abstract
As a step toward gaining a better understanding of the physicochemical bases of pK(a)-value shifts in ion channels, we have previously proposed a method for estimating the proton affinities of systematically engineered ionizable side chains from the kinetic analysis of single-channel current recordings. We reported that the open-channel current flowing through mutants of the (cation-selective) muscle nicotinic acetylcholine receptor (AChR) engineered to bear single basic residues in the transmembrane portion of the pore domain fluctuates between two levels of conductance. Our observations were consistent with the idea that these fluctuations track directly the alternate protonation-deprotonation of basic side chains: protonation of the introduced basic group would attenuate the single-channel conductance, whereas its deprotonation would restore the wild-type-like level. Thus, analysis of the kinetics of these transitions was interpreted to yield the pK(a) values of the substituted side chains. However, other mechanisms can be postulated that would also be consistent with some of our findings but according to which the kinetic analysis of the fluctuations would not yield true pK(a)s. Such mechanisms include the pH-dependent interconversion between two conformations of the channel that, while both ion permeable, would support different cation-conduction rates. In this article, we present experimental evidence for the notion that the fluctuations of the open-channel current observed for the muscle AChR result from the electrostatic interaction between fixed charges and the passing cations rather than from a change in conformation. Hence, we conclude that bona fide pK(a) values can be obtained from single-channel recordings.
Copyright © 2011 Wiley-Liss, Inc.
Figures
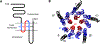


References
-
- Perutz MF. Electrostatic effects in proteins. Science 1978;201:1187–1191. - PubMed
-
- Warshel A Calculations of enzymatic reactions: calculations of pKa, proton transfer reactions, and general catalysis reactions in enzymes. Biochemistry 1981;20:3167–3177. - PubMed
-
- Davis ME, McCammon JA. Electrostatics in biomolecular structure and dynamics. Chem Rev 1990;90:509–521.
-
- Honig B, Nicholls A. Classical electrostatics in biology and chemistry. Science 1995;268:144–1149. - PubMed
-
- García-Moreno EB, Dwyer JJ, Gittis AG, Lattman EE, Spencer DS, Stites WE. Experimental measurement of the effective dielectric in the hydrophobic core of a protein. Biophys Chem 1997;64:211–224. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

