Tumor-specific silencing of COPZ2 gene encoding coatomer protein complex subunit ζ 2 renders tumor cells dependent on its paralogous gene COPZ1
- PMID: 21746916
- PMCID: PMC3145676
- DOI: 10.1073/pnas.1103842108
Tumor-specific silencing of COPZ2 gene encoding coatomer protein complex subunit ζ 2 renders tumor cells dependent on its paralogous gene COPZ1
Abstract
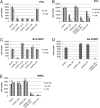
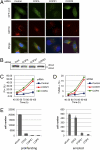
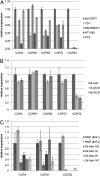
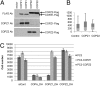
Anticancer drugs are effective against tumors that depend on the molecular target of the drug. Known targets of cytotoxic anticancer drugs are involved in cell proliferation; drugs acting on such targets are ineffective against nonproliferating tumor cells, survival of which leads to eventual therapy failure. Function-based genomic screening identified the coatomer protein complex ζ1 (COPZ1) gene as essential for different tumor cell types but not for normal cells. COPZ1 encodes a subunit of coatomer protein complex 1 (COPI) involved in intracellular traffic and autophagy. The knockdown of COPZ1, but not of COPZ2 encoding isoform coatomer protein complex ζ2, caused Golgi apparatus collapse, blocked autophagy, and induced apoptosis in both proliferating and nondividing tumor cells. In contrast, inhibition of normal cell growth required simultaneous knockdown of both COPZ1 and COPZ2. COPZ2 (but not COPZ1) was down-regulated in the majority of tumor cell lines and in clinical samples of different cancer types. Reexpression of COPZ2 protected tumor cells from killing by COPZ1 knockdown, indicating that tumor cell dependence on COPZ1 is the result of COPZ2 silencing. COPZ2 displays no tumor-suppressive activities, but it harbors microRNA 152, which is silenced in tumor cells concurrently with COPZ2 and acts as a tumor suppressor in vitro and in vivo. Silencing of microRNA 152 in different cancers and the ensuing down-regulation of its host gene COPZ2 offer a therapeutic opportunity for proliferation-independent selective killing of tumor cells by COPZ1-targeting agents.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Loss of COPZ1 induces NCOA4 mediated autophagy and ferroptosis in glioblastoma cell lines.Oncogene. 2021 Feb;40(8):1425-1439. doi: 10.1038/s41388-020-01622-3. Epub 2021 Jan 8. Oncogene. 2021. PMID: 33420375 Free PMC article.
-
Targeting COPZ1 non-oncogene addiction counteracts the viability of thyroid tumor cells.Cancer Lett. 2017 Dec 1;410:201-211. doi: 10.1016/j.canlet.2017.09.024. Epub 2017 Sep 23. Cancer Lett. 2017. PMID: 28951131
-
COPZ1 regulates ferroptosis through NCOA4-mediated ferritinophagy in lung adenocarcinoma.Biochim Biophys Acta Gen Subj. 2024 Nov;1868(11):130706. doi: 10.1016/j.bbagen.2024.130706. Epub 2024 Aug 23. Biochim Biophys Acta Gen Subj. 2024. PMID: 39181476
-
COPB2: a transport protein with multifaceted roles in cancer development and progression.Clin Transl Oncol. 2021 Nov;23(11):2195-2205. doi: 10.1007/s12094-021-02630-9. Epub 2021 Jun 8. Clin Transl Oncol. 2021. PMID: 34101128 Free PMC article. Review.
-
[RNA interference: biogenesis molecular mechanisms and its applications in cervical cancer].Rev Invest Clin. 2010 Jan-Feb;62(1):63-80. Rev Invest Clin. 2010. PMID: 20415061 Review. Spanish.
Cited by
-
Identification and validation of a novel signature for prediction the prognosis and immunotherapy benefit in bladder cancer.PeerJ. 2022 Jan 25;10:e12843. doi: 10.7717/peerj.12843. eCollection 2022. PeerJ. 2022. PMID: 35127296 Free PMC article.
-
Loss of COPZ1 induces NCOA4 mediated autophagy and ferroptosis in glioblastoma cell lines.Oncogene. 2021 Feb;40(8):1425-1439. doi: 10.1038/s41388-020-01622-3. Epub 2021 Jan 8. Oncogene. 2021. PMID: 33420375 Free PMC article.
-
Regulation of colony stimulating factor-1 expression and ovarian cancer cell behavior in vitro by miR-128 and miR-152.Mol Cancer. 2012 Aug 21;11:58. doi: 10.1186/1476-4598-11-58. Mol Cancer. 2012. PMID: 22909061 Free PMC article.
-
Towards unraveling the human tooth transcriptome: the dentome.PLoS One. 2015 Apr 7;10(4):e0124801. doi: 10.1371/journal.pone.0124801. eCollection 2015. PLoS One. 2015. PMID: 25849153 Free PMC article.
-
Coding and regulatory somatic profiling of triple-negative breast cancer in Sub-Saharan African patients.Sci Rep. 2025 Mar 25;15(1):10325. doi: 10.1038/s41598-025-94707-6. Sci Rep. 2025. PMID: 40133516 Free PMC article.
References
-
- Garnock-Jones KP, Keating GM, Scott LJ. Trastuzumab: A review of its use as adjuvant treatment in human epidermal growth factor receptor 2 (HER2)-positive early breast cancer. Drugs. 2010;70:215–239. - PubMed
-
- Yoshida T, Zhang G, Haura EB. Targeting epidermal growth factor receptor: Central signaling kinase in lung cancer. Biochem Pharmacol. 2010;80:613–623. - PubMed
-
- Collins SJ. Retinoic acid receptors, hematopoiesis and leukemogenesis. Curr Opin Hematol. 2008;15:346–351. - PubMed
-
- Weinstein IB, Joe A. Oncogene addiction. Cancer Res. 2008;68:3077–3080, discussion 3080. - PubMed
-
- Primiano T, et al. Identification of potential anticancer drug targets through the selection of growth-inhibitory genetic suppressor elements. Cancer Cell. 2003;4:41–53. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

