CD39/adenosine pathway is involved in AIDS progression
- PMID: 21750674
- PMCID: PMC3131268
- DOI: 10.1371/journal.ppat.1002110
CD39/adenosine pathway is involved in AIDS progression
Abstract
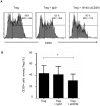
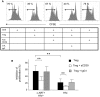
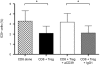
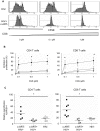
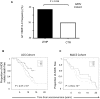
HIV-1 infection is characterized by a chronic activation of the immune system and suppressed function of T lymphocytes. Regulatory CD4+ CD25(high) FoxP3+CD127(low) T cells (Treg) play a key role in both conditions. Here, we show that HIV-1 positive patients have a significant increase of Treg-associated expression of CD39/ENTPD1, an ectoenzyme which in concert with CD73 generates adenosine. We show in vitro that the CD39/adenosine axis is involved in Treg suppression in HIV infection. Treg inhibitory effects are relieved by CD39 down modulation and are reproduced by an adenosine-agonist in accordance with a higher expression of the adenosine A2A receptor on patients' T cells. Notably, the expansion of the Treg CD39+ correlates with the level of immune activation and lower CD4+ counts in HIV-1 infected patients. Finally, in a genetic association study performed in three different cohorts, we identified a CD39 gene polymorphism that was associated with down-modulated CD39 expression and a slower progression to AIDS.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures


References
-
- Cabrera R, Tu Z, Xu Y, Firpi RJ, Rosen HR, et al. An immunomodulatory role for CD4(+)CD25(+) regulatory T lymphocytes in hepatitis C virus infection. Hepatology. 2004;40:1062–1071. - PubMed
-
- Kinter AL, Hennessey M, Bell A, Kern S, Lin Y, et al. CD25(+)CD4(+) regulatory T cells from the peripheral blood of asymptomatic HIV-infected individuals regulate CD4(+) and CD8(+) HIV-specific T cell immune responses in vitro and are associated with favorable clinical markers of disease status. J Exp Med. 2004;200:331–343. - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

