Increased fitness of rice plants to abiotic stress via habitat adapted symbiosis: a strategy for mitigating impacts of climate change
- PMID: 21750695
- PMCID: PMC3130040
- DOI: 10.1371/journal.pone.0014823
Increased fitness of rice plants to abiotic stress via habitat adapted symbiosis: a strategy for mitigating impacts of climate change
Abstract
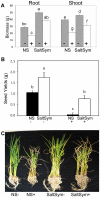
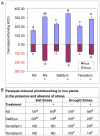
Climate change and catastrophic events have contributed to rice shortages in several regions due to decreased water availability and soil salinization. Although not adapted to salt or drought stress, two commercial rice varieties achieved tolerance to these stresses by colonizing them with Class 2 fungal endophytes isolated from plants growing across moisture and salinity gradients.Plant growth and development, water usage, ROS sensitivity and osmolytes were measured with and without stress under controlled conditions.The endophytes conferred salt, drought and cold tolerance to growth chamber and greenhouse grown plants. Endophytes reduced water consumption by 20-30% and increased growth rate, reproductive yield, and biomass of greenhouse grown plants. In the absence of stress, there was no apparent cost of the endophytes to plants, however, endophyte colonization decreased from 100% at planting to 65% compared to greenhouse plants grown under continual stress (maintained 100% colonization).These findings indicate that rice plants can exhibit enhanced stress tolerance via symbiosis with Class 2 endophytes, and suggest that symbiotic technology may be useful in mitigating impacts of climate change on other crops and expanding agricultural production onto marginal lands.
Conflict of interest statement
Figures



References
-
- Bertin RI. Plant phenology and distribution in relation to recent climate change. Journal of the Torrey Botanical Society. 2008;135:126–146.
-
- Pianka ER. Latitudinal gradients in species diversity - a review of concepts. American Naturalist. 1966;100:33–46.
-
- Stevens GC. The latitudinal gradient in geographical range - how so many species coexist in the tropics. American Naturalist. 1989;133:240–256.
-
- Bradshaw AD. Evolutionary significance of phenotypic plasticity in plants. Advances in Genetics. 1965;13:115–155.
-
- Givnish TJ. Ecological constraints on the evolution of plasticity in plants. Evolutionary Ecology. 2002;16:213–242.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

