Total synthesis of (+)-gliocladin C enabled by visible-light photoredox catalysis
- PMID: 21751318
- PMCID: PMC3496252
- DOI: 10.1002/anie.201103145
Total synthesis of (+)-gliocladin C enabled by visible-light photoredox catalysis
Figures


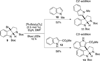
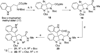

References
-
-
a) Bisindole Alkaloids“: Cordell GA, Saxton JE. In: The Alkaloids: Chemistry and Physiology. Manske RHF, Rodrigo RGA, editors. Vol. 20. New York: Academic Press; 1981. pp. 3–294. b) ”Chemistry and Reactions of Cyclic Tautomers of Tryptamines and Tryptophans“: Hino T, Nakagawa M. In: The Alkaloids: Chemistry and Pharmacology. Brossi A, editor. Vol. 34. New York: Academic Press; 1989. pp. 1–75. c) ”Alkaloids from the Medicinal Plants of New Caledonia“: Sévenet T, Pusset J. In: The Alkaloids: Chemistry and Pharmacology. Cordell GA, editor. Vol. 48. New York: Academic Press; 1996. pp. 58–59.
-
-
- Usami Y, Yamaguchi J, Numata A. Heterocycles. 2004;63:1123.
-
- Dong J-Y, He H-P, Shen Y-M, Zhang K-Q. J. Nat. Prod. 2005;68:1510. - PubMed
-
- Takahashi C, Numata A, Matsumura E, Minoura K, Eto H, Shingu T, Ito T, Hasegawa T. J. Antibiot. 1994;47:1242. - PubMed
-
- Zheng CJ, Kim CJ, Bae KS, Kim YH, Kim WG. J. Nat. Prod. 2006;69:1816. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

