A mechanism-based potent sirtuin inhibitor containing Nε-thiocarbamoyl-lysine (TuAcK)
- PMID: 21752644
- PMCID: PMC3146582
- DOI: 10.1016/j.bmcl.2011.06.069
A mechanism-based potent sirtuin inhibitor containing Nε-thiocarbamoyl-lysine (TuAcK)
Abstract
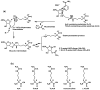
In the current study, we have identified N(ε)-thiocarbamoyl-lysine (TuAcK) as a general sirtuin inhibitory warhead which was shown to be able to confer potent sirtuin inhibition. This inhibition was also shown to be mechanism-based in that the TuAck residue was able to be processed by a sirtuin enzyme with the formation of a stalled S-alkylamidate intermediate.
Copyright © 2011 Elsevier Ltd. All rights reserved.
Figures



Similar articles
-
Simple N(ε)-thioacetyl-lysine-containing cyclic peptides exhibiting highly potent sirtuin inhibition.Bioorg Med Chem Lett. 2016 Mar 15;26(6):1612-1617. doi: 10.1016/j.bmcl.2016.01.086. Epub 2016 Feb 1. Bioorg Med Chem Lett. 2016. PMID: 26874402
-
The mimics of Nε-acyl-lysine derived from cysteine as sirtuin inhibitors.Bioorg Med Chem Lett. 2018 Aug 1;28(14):2375-2378. doi: 10.1016/j.bmcl.2018.06.030. Epub 2018 Jun 18. Bioorg Med Chem Lett. 2018. PMID: 29921476
-
Novel thiourea-based sirtuin inhibitory warheads.Bioorg Med Chem Lett. 2015 Aug 15;25(16):3319-24. doi: 10.1016/j.bmcl.2015.05.058. Epub 2015 May 30. Bioorg Med Chem Lett. 2015. PMID: 26081291 Free PMC article.
-
Sirtuin Inhibition: Strategies, Inhibitors, and Therapeutic Potential.Trends Pharmacol Sci. 2017 May;38(5):459-472. doi: 10.1016/j.tips.2017.01.009. Epub 2017 Apr 5. Trends Pharmacol Sci. 2017. PMID: 28389129 Review.
-
Sirtuin mechanism and inhibition: explored with N(ε)-acetyl-lysine analogs.Mol Biosyst. 2011 Jan;7(1):16-28. doi: 10.1039/c0mb00033g. Epub 2010 Sep 15. Mol Biosyst. 2011. PMID: 20842312 Review.
Cited by
-
Novel Lysine-Based Thioureas as Mechanism-Based Inhibitors of Sirtuin 2 (SIRT2) with Anticancer Activity in a Colorectal Cancer Murine Model.J Med Chem. 2019 Apr 25;62(8):4131-4141. doi: 10.1021/acs.jmedchem.9b00191. Epub 2019 Apr 15. J Med Chem. 2019. PMID: 30986062 Free PMC article.
-
Current Trends in Sirtuin Activator and Inhibitor Development.Molecules. 2024 Mar 6;29(5):1185. doi: 10.3390/molecules29051185. Molecules. 2024. PMID: 38474697 Free PMC article. Review.
-
Structural basis of SIRT7 nucleosome engagement and substrate specificity.Nat Commun. 2025 Feb 4;16(1):1328. doi: 10.1038/s41467-025-56529-y. Nat Commun. 2025. PMID: 39900593 Free PMC article.
-
Isoxazole-Derived Amino Acids are Bromodomain-Binding Acetyl-Lysine Mimics: Incorporation into Histone H4 Peptides and Histone H3.Angew Chem Int Ed Engl. 2016 Jul 11;55(29):8353-7. doi: 10.1002/anie.201602908. Epub 2016 Jun 6. Angew Chem Int Ed Engl. 2016. PMID: 27264992 Free PMC article.
-
Structural Basis of Sirtuin 6-Catalyzed Nucleosome Deacetylation.J Am Chem Soc. 2023 Mar 29;145(12):6811-6822. doi: 10.1021/jacs.2c13512. Epub 2023 Mar 17. J Am Chem Soc. 2023. PMID: 36930461 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Chemical Information

