IFN-β mediates suppression of IL-12p40 in human dendritic cells following infection with virulent Francisella tularensis
- PMID: 21753150
- PMCID: PMC3150225
- DOI: 10.4049/jimmunol.1100377
IFN-β mediates suppression of IL-12p40 in human dendritic cells following infection with virulent Francisella tularensis
Abstract
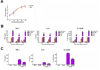
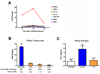
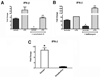
Active suppression of inflammation is a strategy used by many viral and bacterial pathogens, including virulent strains of the bacterium Francisella tularensis, to enable colonization and infection in susceptible hosts. In this study, we demonstrated that virulent F. tularensis strain SchuS4 selectively inhibits production of IL-12p40 in primary human cells via induction of IFN-β. In contrast to the attenuated live vaccine strain, infection of human dendritic cells with virulent SchuS4 failed to induce production of many cytokines associated with inflammation (e.g., TNF-α and IL-12p40). Furthermore, SchuS4 actively suppressed secretion of these cytokines. Assessment of changes in the expression of host genes associated with suppression of inflammatory responses revealed that SchuS4, but not live vaccine strain, induced IFN-β following infection of human dendritic cells. Phagocytosis of SchuS4 and endosomal acidification were required for induction of IFN-β. Further, using a defined mutant of SchuS4, we demonstrated that the presence of bacteria in the cytosol was required, but not sufficient, for induction of IFN-β. Surprisingly, unlike previous reports, induction of IFN-β by F. tularensis was not required for activation of the inflammasome, was not associated with exacerbation of inflammatory responses, and did not control SchuS4 replication when added exogenously. Rather, IFN-β selectively suppressed the ability of SchuS4-infected dendritic cells to produce IL-12p40. Together, these data demonstrated a novel mechanism by which virulent bacteria, in contrast to attenuated strains, modulate human cells to cause disease.
Figures


References
-
- Bosio CM, Aman MJ, Grogan C, Hogan R, Ruthel G, Negley D, Mohamadzadeh M, Bavari S, Schmaljohn A. Ebola and Marburg viruses replicate in monocyte-derived dendritic cells without inducing the production of cytokines and full maturation. J Infect Dis. 2003;188:1630–1638. - PubMed
-
- Bosio CM, Bielefeldt-Ohmann H, Belisle JT. Active suppression of the pulmonary immune response by Francisella tularensis Schu4. J Immunol. 2007;178:4538–4547. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

