N'-[1-(2,4-Dioxo-3,4-dihydro-2H-1-benzopyran-3-yl-idene)eth-yl]thiophene-2-carbo-hydrazide
- PMID: 21754031
- PMCID: PMC3099777
- DOI: 10.1107/S1600536811010907
N'-[1-(2,4-Dioxo-3,4-dihydro-2H-1-benzopyran-3-yl-idene)eth-yl]thiophene-2-carbo-hydrazide
Abstract
The title compound, C(16)H(12)N(2)O(4)S, was obtained by the condensation of 3-acetyl-4-hy-droxy-coumarin with thien-2-ylcarbonyl hydrazide. The pyran ring adopts a 2,4-dione tautomeric form. The benzopyran ring system is almost coplanar with the thio-phene ring [dihedral angle 0.9 (2)°]. The exocyclic C=C double bond has an E geometry. The mol-ecular conformation is stabilized by an intra-molecular N-H⋯O hydrogen bond. In the crystal, inter-molecular N-H⋯O hydrogen bonds link the mol-ecules into chains along the a axis.
Figures

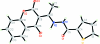

References
-
- Bruker (2001). SMART Bruker AXS Inc., Madison, Wisconsin, USA.
-
- Bruker (2002). SAINT Bruker AXS Inc., Madison, Wisconsin, USA.
-
- Burla, M. C., Caliandro, R., Camalli, M., Carrozzini, B., Cascarano, G. L., De Caro, L., Giacovazzo, C., Polidori, G. & Spagna, R. (2005). J. Appl. Cryst. 38, 381–388.
-
- Kotali, A. (2009). Arkivoc, i, 81–96.
-
- Kotali, A., Kotali, E., Lafazanis, I. S. & Harris, P. A. (2010). Curr. Org. Synth. 7, 62–77.
LinkOut - more resources
Full Text Sources
