Probabilistic tractography recovers a rostrocaudal trajectory of connectivity variability in the human insular cortex
- PMID: 21761507
- PMCID: PMC3443376
- DOI: 10.1002/hbm.21338
Probabilistic tractography recovers a rostrocaudal trajectory of connectivity variability in the human insular cortex
Abstract
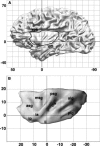
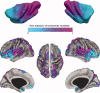
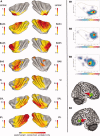
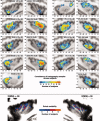
The insular cortex of macaques has a wide spectrum of anatomical connections whose distribution is related to its heterogeneous cytoarchitecture. Although there is evidence of a similar cytoarchitectural arrangement in humans, the anatomical connectivity of the insula in the human brain has not yet been investigated in vivo. In the present work, we used in vivo probabilistic white-matter tractography and Laplacian eigenmaps (LE) to study the variation of connectivity patterns across insular territories in humans. In each subject and hemisphere, we recovered a rostrocaudal trajectory of connectivity variation ranging from the anterior dorsal and ventral insula to the dorsal caudal part of the long insular gyri. LE suggested that regional transitions among tractography patterns in the insula occur more gradually than in other brain regions. In particular, the change in tractography patterns was more gradual in the insula than in the medial premotor region, where a sharp transition between different tractography patterns was found. The recovered trajectory of connectivity variation in the insula suggests a relation between connectivity and cytoarchitecture in humans resembling that previously found in macaques: tractography seeds from the anterior insula were mainly found in limbic and paralimbic regions and in anterior parts of the inferior frontal gyrus, while seeds from caudal insular territories mostly reached parietal and posterior temporal cortices. Regions in the putative dysgranular insula displayed more heterogeneous connectivity patterns, with regional differences related to the proximity with either putative granular or agranular regions.
Copyright © 2011 Wiley Periodicals, Inc.
Figures




Similar articles
-
Probabilistic fibre tract analysis of cytoarchitectonically defined human inferior parietal lobule areas reveals similarities to macaques.Neuroimage. 2011 Sep 15;58(2):362-80. doi: 10.1016/j.neuroimage.2011.06.027. Epub 2011 Jun 21. Neuroimage. 2011. PMID: 21718787 Free PMC article.
-
Functional connectivity of the insula in the resting brain.Neuroimage. 2011 Mar 1;55(1):8-23. doi: 10.1016/j.neuroimage.2010.11.049. Epub 2010 Nov 24. Neuroimage. 2011. PMID: 21111053
-
Structural Connectivity Differences Reflect Microstructural Heterogeneity of the Human Insular Cortex.Hum Brain Mapp. 2025 Jun 1;46(8):e70231. doi: 10.1002/hbm.70231. Hum Brain Mapp. 2025. PMID: 40396764 Free PMC article.
-
Circuitry and functional aspects of the insular lobe in primates including humans.Brain Res Brain Res Rev. 1996 Oct;22(3):229-44. doi: 10.1016/s0165-0173(96)00011-2. Brain Res Brain Res Rev. 1996. PMID: 8957561 Review.
-
The insular cortex: a review.Prog Brain Res. 2012;195:123-63. doi: 10.1016/B978-0-444-53860-4.00007-6. Prog Brain Res. 2012. PMID: 22230626 Review.
Cited by
-
Distinct Aging Effects on Functional Networks in Good and Poor Cognitive Performers.Front Aging Neurosci. 2016 Sep 9;8:215. doi: 10.3389/fnagi.2016.00215. eCollection 2016. Front Aging Neurosci. 2016. PMID: 27667972 Free PMC article.
-
Aberrant Brain Functional Connectivity Dynamic Responses to the Valsalva Maneuver in Heart Failure.J Card Fail. 2019 Sep;25(9):757-766. doi: 10.1016/j.cardfail.2019.06.010. Epub 2019 Jun 29. J Card Fail. 2019. PMID: 31265919 Free PMC article.
-
Dynamic functional network connectivity reveals unique and overlapping profiles of insula subdivisions.Hum Brain Mapp. 2016 May;37(5):1770-87. doi: 10.1002/hbm.23135. Epub 2016 Feb 16. Hum Brain Mapp. 2016. PMID: 26880689 Free PMC article.
-
Differential structural and resting state connectivity between insular subdivisions and other pain-related brain regions.Pain. 2014 Oct;155(10):2047-55. doi: 10.1016/j.pain.2014.07.009. Epub 2014 Jul 15. Pain. 2014. PMID: 25047781 Free PMC article.
-
Neural pathways of embarrassment and their modulation by social anxiety.Neuroimage. 2015 Oct 1;119:252-261. doi: 10.1016/j.neuroimage.2015.06.036. Epub 2015 Jun 18. Neuroimage. 2015. PMID: 26093329 Free PMC article.
References
-
- Ackermann H, Riecker A ( 2004): The contribution of the insula to motor aspects of speech production: A review and a hypothesis. Brain Lang 89: 320–328. - PubMed
-
- Ackermann H, Riecker A ( 2010): The contribution(s) of the insula to speech production: A review of the clinical and functional imaging literature. Brain Struct Funct 214: 419–433. - PubMed
-
- Ackermann H, Riecker A, Mathiak K, Erb M, Grodd W, Wildgruber D ( 2001): Rate‐dependent activation of a prefrontal‐insular‐cerebellar network during passive listening to trains of click stimuli: An fMRI study. Neuroreport 12: 4087–4092. - PubMed
-
- Afif A, Mertens P ( 2010): Description of sulcal organization of the insular cortex. Surg Radiol Anat 32: 491–498. - PubMed
-
- Aggleton JP, Burton MJ, Passingham RE ( 1980): Cortical and subcortical afferents to the amygdala of the rhesus monkey (Macaca mulatta). Brain Res 190: 347–368. - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

