Human papillomavirus type 18 chimeras containing the L2/L1 capsid genes from evolutionarily diverse papillomavirus types generate infectious virus
- PMID: 21762735
- PMCID: PMC3163805
- DOI: 10.1016/j.virusres.2011.06.024
Human papillomavirus type 18 chimeras containing the L2/L1 capsid genes from evolutionarily diverse papillomavirus types generate infectious virus
Abstract
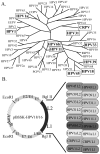
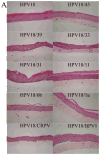
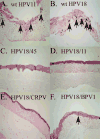
Papillomaviruses (PVs) comprise a large family of viruses infecting nearly all vertebrate species, with more than 100 human PVs identified. Our previous studies showed that a mutant chimera HPV18/16 genome, consisting of the upper regulatory region and early ORFs of HPV18 and the late ORFs of HPV16, was capable of producing infectious virus in organotypic raft cultures. We were interested in determining whether the ability of this chimeric genome to produce infectious virus was the result of HPV18 and HPV16 being similarly oncogenic, anogenital types and whether more disparate PV types could also interact functionally. To test this we created a series of HPV18 chimeric genomes where the ORFs for the HPV18 capsid genes were replaced with the capsid genes of HPV45, HPV39, HPV33, HPV31, HPV11, HPV6b, HPV1a, CRPV, and BPV1. All chimeras were able to produce infectious chimeric viral particles, although with lower infectivity than wild-type HPV18. Steps in the viral life cycle and characteristics of the viral particles were examined to identify potential causes for the decrease in infectivity.
Copyright © 2011 Elsevier B.V. All rights reserved.
Figures



Similar articles
-
Study of infectious virus production from HPV18/16 capsid chimeras.Virology. 2010 Sep 30;405(2):289-99. doi: 10.1016/j.virol.2010.05.019. Epub 2010 Jul 3. Virology. 2010. PMID: 20598725 Free PMC article.
-
Infectious virions produced from a human papillomavirus type 18/16 genomic DNA chimera.J Virol. 2002 May;76(10):4723-33. doi: 10.1128/jvi.76.10.4723-4733.2002. J Virol. 2002. PMID: 11967289 Free PMC article.
-
L1 interaction domains of papillomavirus l2 necessary for viral genome encapsidation.J Virol. 2001 May;75(9):4332-42. doi: 10.1128/JVI.75.9.4332-4342.2001. J Virol. 2001. PMID: 11287582 Free PMC article.
-
Worldwide genetic variations in high-risk human papillomaviruses capsid L1 gene and their impact on vaccine efficiency.Gene. 2021 May 25;782:145533. doi: 10.1016/j.gene.2021.145533. Epub 2021 Feb 23. Gene. 2021. PMID: 33636291 Review.
-
Developments in L2-based human papillomavirus (HPV) vaccines.Virus Res. 2017 Mar 2;231:166-175. doi: 10.1016/j.virusres.2016.11.020. Epub 2016 Nov 23. Virus Res. 2017. PMID: 27889616 Free PMC article. Review.
Cited by
-
Superinfection Exclusion between Two High-Risk Human Papillomavirus Types during a Coinfection.J Virol. 2018 Mar 28;92(8):e01993-17. doi: 10.1128/JVI.01993-17. Print 2018 Apr 15. J Virol. 2018. PMID: 29437958 Free PMC article.
-
Three-dimensional cell culture models for investigating human viruses.Virol Sin. 2016 Oct;31(5):363-379. doi: 10.1007/s12250-016-3889-z. Epub 2016 Oct 27. Virol Sin. 2016. PMID: 27822716 Free PMC article. Review.
-
Papillomavirus Infectious Pathways: A Comparison of Systems.Viruses. 2015 Aug 4;7(8):4303-25. doi: 10.3390/v7082823. Viruses. 2015. PMID: 26247955 Free PMC article. Review.
References
-
- Becker KA, Florin L, Sapp C, Sapp M. Dissection of human papillomavirus type 33 L2 domains involved in nuclear domains (ND) 10 homing and reorganization. Virology. 2003;314(1):161–7. - PubMed
-
- Bernard HU, Calleja-Macias IE, Dunn ST. Genome variation of human papillomavirus types: phylogenetic and medical implications. Int J Cancer. 2006;118(5):1071–6. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

