The cellular basis for animal regeneration
- PMID: 21763617
- PMCID: PMC3139400
- DOI: 10.1016/j.devcel.2011.06.016
The cellular basis for animal regeneration
Abstract
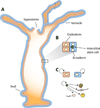
The ability of animals to regenerate missing parts is a dramatic and poorly understood aspect of biology. The sources of new cells for these regenerative phenomena have been sought for decades. Recent advances involving cell fate tracking in complex tissues have shed new light on the cellular underpinnings of regeneration in Hydra, planarians, zebrafish, Xenopus, and Axolotl. Planarians accomplish regeneration with use of adult pluripotent stem cells, whereas several vertebrates utilize a collection of lineage-restricted progenitors from different tissues. Together, an array of cellular strategies-from pluripotent stem cells to tissue-specific stem cells and dedifferentiation-are utilized for regeneration.
Copyright © 2011 Elsevier Inc. All rights reserved.
Figures
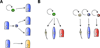



References
-
- Aboobaker AA. Planarian stem cells: a simple paradigm for regeneration. Trends Cell Biol. 2011 - PubMed
-
- Adell T, Saló E, Boutros M, Bartscherer K. Smed-Evi/Wntless is required for beta-catenin-dependent and -independent processes during planarian regeneration. Development. 2009;136:905–910. - PubMed
-
- Akimenko MA, Mari-Beffa M, Becerra J, Geraudie J. Old questions, new tools, and some answers to the mystery of fin regeneration. Dev Dyn. 2003;226:190–201. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

