Demonstration and Characterization of Biomolecular Enrichment on Microfluidic Aptamer-Functionalized Surfaces
- PMID: 21765612
- PMCID: PMC3135969
- DOI: 10.1016/j.snb.2010.11.024
Demonstration and Characterization of Biomolecular Enrichment on Microfluidic Aptamer-Functionalized Surfaces
Abstract
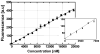
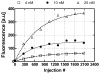
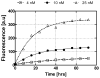
This paper demonstrates and systematically characterizes the enrichment of biomolecular compounds using aptamer-functionalized surfaces within a microfluidic device. The device consists of a microchamber packed with aptamer-functionalized microbeads and integrated with a microheater and temperature sensor to enable thermally controlled binding and release of biomolecules by the aptamer. We first present an equilibrium binding-based analytical model to understand the enrichment process. The characteristics of the aptamer-analyte binding and enrichment are then experimentally studied, using adenosine monophosphate (AMP) and a specific RNA aptamer as a model system. The temporal process of AMP binding to the aptamer is found to be primarily determined by the aptamer-AMP binding kinetics. The temporal process of aptamer-AMP dissociation at varying temperatures is also obtained and observed to occur relatively rapidly (< 2 s). The specificity of the enrichment is next confirmed by performing selective enrichment of AMP from a sample containing biomolecular impurities. Finally, we investigate the enrichment of AMP by either discrete or continuous introduction of a dilute sample into the microchamber, demonstrating enrichment factors ranging from 566 to 686×, which agree with predictions of the analytical model.
Figures


Similar articles
-
Specific capture and temperature-mediated release of cells in an aptamer-based microfluidic device.Lab Chip. 2012 Sep 21;12(18):3504-13. doi: 10.1039/c2lc40411g. Epub 2012 Aug 2. Lab Chip. 2012. PMID: 22854859 Free PMC article.
-
Microcantilever-Based Label-Free Characterization of Temperature-Dependent Biomolecular Affinity Binding.Sens Actuators B Chem. 2013 Jan 1;176:653-659. doi: 10.1016/j.snb.2012.02.045. Sens Actuators B Chem. 2013. PMID: 24723743 Free PMC article.
-
Aptamer-based microfluidic beads array sensor for simultaneous detection of multiple analytes employing multienzyme-linked nanoparticle amplification and quantum dots labels.Biosens Bioelectron. 2014 Jul 15;57:22-9. doi: 10.1016/j.bios.2014.01.054. Epub 2014 Feb 5. Biosens Bioelectron. 2014. PMID: 24534576
-
Aptamer selection via versatile microfluidic platforms and their diverse applications.Lab Chip. 2025 Feb 25;25(5):1047-1080. doi: 10.1039/d4lc00859f. Lab Chip. 2025. PMID: 39774569 Review.
-
Screening of aptamers on microfluidic systems for clinical applications.Sensors (Basel). 2012;12(7):9514-29. doi: 10.3390/s120709514. Epub 2012 Jul 11. Sensors (Basel). 2012. PMID: 23012556 Free PMC article. Review.
Cited by
-
Nucleic Acid Isolation and Enrichment on a Microchip.Sens Actuators A Phys. 2013 Jun 1;195:183-190. doi: 10.1016/j.sna.2012.07.022. Sens Actuators A Phys. 2013. PMID: 24729660 Free PMC article.
-
Advances in microfluidic materials, functions, integration, and applications.Chem Rev. 2013 Apr 10;113(4):2550-83. doi: 10.1021/cr300337x. Epub 2013 Feb 14. Chem Rev. 2013. PMID: 23410114 Free PMC article. Review. No abstract available.
-
Integrated Microfluidic Aptasensor for Mass Spectrometric Detection of Vasopressin in Human Plasma Ultrafiltrate.Anal Methods. 2016 Jul 14;8(26):5190-5196. doi: 10.1039/C5AY02979A. Epub 2016 May 11. Anal Methods. 2016. PMID: 28090219 Free PMC article.
-
Specific capture and temperature-mediated release of cells in an aptamer-based microfluidic device.Lab Chip. 2012 Sep 21;12(18):3504-13. doi: 10.1039/c2lc40411g. Epub 2012 Aug 2. Lab Chip. 2012. PMID: 22854859 Free PMC article.
-
Spatially selective release of aptamer-captured cells by temperature mediation.IET Nanobiotechnol. 2014 Mar;8(1):2-9. doi: 10.1049/iet-nbt.2013.0028. IET Nanobiotechnol. 2014. PMID: 24888185 Free PMC article.
References
-
- Rios A, Escarpa A, Gonzalez MC, Crevillen AG. Challenges of Analytical Microsysterns. Trac-Trends in Analytical Chemistry. 2006;25:467–479.
-
- Crevillen AG, Hervas M, Lopez MA, Gonzalez MC, Escarpa A. Real Sample Analysis on Microfluidic Devices. Talanta. 2007;74:342–357. - PubMed
-
- Song S, Singh AK. On-Chip Sample Preconcentration for Integrated Microfluidic Analysis. Analytical and Bioanalytical Chemistry. 2006;384:41–43. - PubMed
-
- Yang Y, Li C, Lee KH, Craighead HG. Coupling on-Chip Solid-Phase Extraction to Electrospray Mass Spectrometry through Integrated Electrospray Tip. Electrophoresis. 2005;26:3622–3630. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
