Cytochromes P450 catalyze the reduction of α,β-unsaturated aldehydes
- PMID: 21766881
- PMCID: PMC3180908
- DOI: 10.1021/tx200080b
Cytochromes P450 catalyze the reduction of α,β-unsaturated aldehydes
Abstract
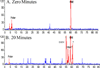
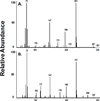
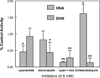
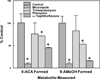
The metabolism of α,β-unsaturated aldehydes, e.g., 4-hydroxynonenal, involves oxidation to carboxylic acids, reduction to alcohols, and glutathionylation to eventually form mercapturide conjugates. Recently, we demonstrated that P450s can oxidize aldehydes to carboxylic acids, a reaction previously thought to involve aldehyde dehydrogenase. When recombinant cytochrome P450 3A4 was incubated with 4-hydroxynonenal, O(2), and NADPH, several products were produced, including 1,4-dihydroxynonene (DHN), 4-hydroxy-2-nonenoic acid (HNA), and an unknown metabolite. Several P450s catalyzed the reduction reaction in the order (human) P450 2B6 ≅ P450 3A4 > P450 1A2 > P450 2J2 > (mouse) P450 2c29. Other P450s did not catalyze the reduction reaction (human P450 2E1 and rabbit P450 2B4). Metabolism by isolated rat hepatocytes showed that HNA formation was inhibited by cyanamide, while DHN formation was not affected. Troleandomycin increased HNA production 1.6-fold while inhibiting DHN formation, suggesting that P450 3A11 is a major enzyme involved in rat hepatic clearance of 4-HNE. A fluorescent assay was developed using 9-anthracenealdehyde to measure both reactions. Feeding mice a diet containing t-butylated hydroxyanisole increased the level of both activities with hepatic microsomal fractions but not proportionally. Miconazole (0.5 mM) was a potent inhibitor of these microsomal reduction reactions, while phenytoin and α-naphthoflavone (both at 0.5 mM) were partial inhibitors, suggesting the role of multiple P450 enzymes. The oxidative metabolism of these aldehydes was inhibited >90% in an Ar or CO atmosphere, while the reductive reactions were not greatly affected. These results suggest that P450s are significant catalysts of the reduction of α,β-unsaturated aldehydes in the liver.
Figures


Similar articles
-
Cytochromes P450 catalyze oxidation of alpha,beta-unsaturated aldehydes.Arch Biochem Biophys. 2007 Aug 15;464(2):187-96. doi: 10.1016/j.abb.2007.05.019. Epub 2007 Jun 8. Arch Biochem Biophys. 2007. PMID: 17599801 Free PMC article.
-
Human enzymes involved in the metabolic activation of the environmental contaminant 3-nitrobenzanthrone: evidence for reductive activation by human NADPH:cytochrome p450 reductase.Cancer Res. 2003 Jun 1;63(11):2752-61. Cancer Res. 2003. PMID: 12782579
-
4-Biphenylaldehyde and 9-anthraldehyde: two fluorescent substrates for determining P450 enzyme activities in rat and human.Xenobiotica. 2003 Jan;33(1):1-11. doi: 10.1080/0049825021000017894. Xenobiotica. 2003. PMID: 12519690
-
Multi-step oxidations catalyzed by cytochrome P450 enzymes: Processive vs. distributive kinetics and the issue of carbonyl oxidation in chemical mechanisms.Arch Biochem Biophys. 2011 Mar 1;507(1):126-34. doi: 10.1016/j.abb.2010.08.017. Epub 2010 Sep 4. Arch Biochem Biophys. 2011. PMID: 20804723 Free PMC article. Review.
-
Carboxylic acid reductase enzymes (CARs).Curr Opin Chem Biol. 2018 Apr;43:23-29. doi: 10.1016/j.cbpa.2017.10.006. Epub 2017 Nov 9. Curr Opin Chem Biol. 2018. PMID: 29127833 Review.
Cited by
-
Reactive metabolites in the biotransformation of molecules containing a furan ring.Chem Res Toxicol. 2013 Jan 18;26(1):6-25. doi: 10.1021/tx3003824. Epub 2012 Oct 24. Chem Res Toxicol. 2013. PMID: 23061605 Free PMC article. Review.
-
A targeted proteomics approach for profiling murine cytochrome P450 expression.J Pharmacol Exp Ther. 2014 May;349(2):221-8. doi: 10.1124/jpet.113.212456. Epub 2014 Mar 4. J Pharmacol Exp Ther. 2014. PMID: 24594750 Free PMC article.
-
Protein modification by oxidized phospholipids and hydrolytically released lipid electrophiles: Investigating cellular responses.Biochim Biophys Acta. 2012 Oct;1818(10):2424-35. doi: 10.1016/j.bbamem.2012.04.014. Epub 2012 Apr 27. Biochim Biophys Acta. 2012. PMID: 22562025 Free PMC article. Review.
-
Differential metabolism of 4-hydroxynonenal in liver, lung and brain of mice and rats.Toxicol Appl Pharmacol. 2014 Aug 15;279(1):43-52. doi: 10.1016/j.taap.2014.04.026. Epub 2014 May 14. Toxicol Appl Pharmacol. 2014. PMID: 24832492 Free PMC article.
-
S-adenosyl-l-methionine protection of acetaminophen mediated oxidative stress and identification of hepatic 4-hydroxynonenal protein adducts by mass spectrometry.Toxicol Appl Pharmacol. 2014 Dec 1;281(2):174-84. doi: 10.1016/j.taap.2014.08.027. Epub 2014 Sep 22. Toxicol Appl Pharmacol. 2014. PMID: 25246065 Free PMC article.
References
-
- Nelson TJ, Boor PJ. Allylamine cardiotoxicity--IV. Metabolism to acrolein by cardiovascular tissues. Biochem Pharmacol. 1982 February 15;31(4):509–514. - PubMed
-
- Gardner HW, Dornbos DL, Jr, Desjardins AE. Hexanal, trans-2-hexenal, and trans-2-nonenal inhibit soybean, Glycine max, seed germination. J Agric Food Chem. 1990 June 1;38(6):1316–1320.
-
- Schneider C, Tallman KA, Porter NA, Brash AR. Two distinct pathways of formation of 4-hydroxynonenal. Mechanisms of nonenzymatic transformation of the 9- and 13-hydroperoxides of linoleic acid to 4-hydroxyalkenals. J Biol Chem. 2001 June 15;276(24):20831–20838. - PubMed
-
- Esterbauer H, Schaur RJ, Zollner H. Chemistry and biochemistry of 4-hydroxynonenal, malonaldehyde and related aldehydes. Free Radic Biol Med. 1991;11(1):81–128. - PubMed
-
- Poli G, Schaur RJ. 4-Hydroxynonenal in the pathomechanisms of oxidative stress. IUBMB Life. 2000 October;50(4–5):315–321. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

