A double-blind, randomized, placebo-controlled, active reference study of Lu AA21004 in patients with major depressive disorder
- PMID: 21767441
- PMCID: PMC3349292
- DOI: 10.1017/S1461145711001027
A double-blind, randomized, placebo-controlled, active reference study of Lu AA21004 in patients with major depressive disorder
Abstract
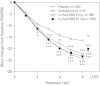
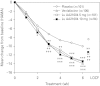
The efficacy, safety, and tolerability of Lu AA21004 vs. placebo using venlafaxine XR as active reference in patients with DSM-IV-TR major depressive disorder (MDD) were evaluated. Lu AA21004 is a novel antidepressant that is a 5-HT3 and 5-HT7 receptor antagonist, 5-HT1A receptor agonist, 5-HT1B receptor partial agonist and inhibitor of the 5-HT transporter in recombinant cell lines. In this 6-wk, multi-site study, 429 patients were randomly assigned (1:1:1:1) to 5 or 10 mg Lu AA21004, placebo or 225 mg venlafaxine XR. All patients had a baseline Montgomery-Åsberg Depression Rating Scale (MADRS) total score ≥ 30. The primary efficacy analysis was based on the MADRS total score adjusting for multiplicity using a hierarchical testing procedure starting with the highest dose vs. placebo. Lu AA21004 was statistically significantly superior to placebo (n=105) in mean change from baseline in MADRS total score at week 6 (p<0.0001, last observation carried forward), with a mean treatment difference vs. placebo of 5.9 (5 mg, n=108), and 5.7 (10 mg, n=100) points. Venlafaxine XR (n=112) was also significantly superior to placebo at week 6 (p<0.0001). In total, 30 patients withdrew due to adverse events (AEs)--placebo: four (4%); 5 mg Lu AA21004: three (3%); 10 mg Lu AA21004: seven (7%); and venlafaxine: 16 (14%). The most common AEs were nausea, headache, hyperhidrosis, and dry mouth. No clinically relevant changes over time were seen in the clinical laboratory results, vital signs, weight, or ECG parameters. In this study, treatment with 5 mg and 10 mg Lu AA21004 for 6 wk was efficacious and well tolerated in patients with MDD.
Trial registration: ClinicalTrials.gov NCT00839423.
Figures

References
-
- Amstislavskaya TG, Popova NK. The roles of different types of serotonin receptors in activation of the hypophyseal-testicular complex induced in mice by the presence of a female. Neuroscience and Behavioral Physiology. 2004;34:833–837. - PubMed
-
- APA. Diagnostic and Statistical Manual of Mental Disorders. 4th edn. Washington, DC: American Psychiatric Association; 1994. Text Revision (DSM-IV-TR).
-
- Areberg J, Dragheim M, Brennum L, Stensbøl TB, San Francisco, CA, USA: 2009. ). Lu AA21004: efficacy at low serotonin transporter occupancy at clinically effective doses – from animal studies to depressed patients. Poster NR7-020 presented at the 162nd Annual Meeting of the American Psychiatric Association, 16–21 May 2009, . Accessed 9 March 2011.
-
- Artigas F, Celada P, Laruelle M, Adell A. How does pindolol improve antidepressant action? Trends in Pharmacological Sciences. 2001;22:224–228. - PubMed
-
- Artigas F, Romero L, de Montigny C, Blier P. Acceleration of the effect of selected antidepressant drugs in major depression by 5-HT1A antagonists. Trends in Neuroscience. 1996;19:378–383. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

