Atomic force microscopy reveals the architecture of the epithelial sodium channel (ENaC)
- PMID: 21775436
- PMCID: PMC3173231
- DOI: 10.1074/jbc.M111.275289
Atomic force microscopy reveals the architecture of the epithelial sodium channel (ENaC)
Abstract
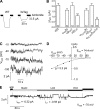
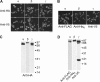
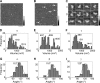
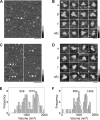
The epithelial sodium channel (ENaC) is a member of the ENaC/degenerin superfamily. ENaC is a heteromultimer containing three homologous subunits (α, β, and γ); however, the subunit stoichiometry is still controversial. Here, we addressed this issue using atomic force microscopy imaging of complexes between isolated ENaC and antibodies/Fab fragments directed against specific epitope tags on the α-, β- and γ-subunits. We show that for α-, β- and γ-ENaC alone, pairs of antibodies decorate the channel at an angle of 120°, indicating that the individual subunits assemble as homotrimers. A similar approach demonstrates that αβγ-ENaC assembles as a heterotrimer containing one copy of each subunit. Intriguingly, all four subunit combinations also produce higher-order structures containing two or three individual trimers. The trimer-of-trimers organization would account for earlier reports that ENaC contains eight to nine subunits.
Figures

References
-
- Kellenberger S., Schild L. (2002) Physiol. Rev. 82, 735–767 - PubMed
-
- Loffing J., Korbmacher C. (2009) Pflügers Arch. 458, 111–135 - PubMed
-
- Canessa C. M., Horisberger J. D., Rossier B. C. (1993) Nature 361, 467–470 - PubMed
-
- Canessa C. M., Schild L., Buell G., Thorens B., Gautschi I., Horisberger J. D., Rossier B. C. (1994) Nature 367, 463–467 - PubMed
-
- Waldmann R., Champigny G., Bassilana F., Voilley N., Lazdunski M. (1995) J. Biol. Chem. 270, 27411–27414 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

