Metastatic breast cancer cells in the bone marrow microenvironment: novel insights into oncoprotection
- PMID: 21776337
- PMCID: PMC3138628
- DOI: 10.1007/s12156-010-0071-y
Metastatic breast cancer cells in the bone marrow microenvironment: novel insights into oncoprotection
Abstract
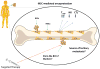
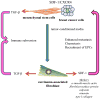
Among all cancers, malignancies of the breast are the second leading cause of cancer death in the United States after carcinoma of the lung. One of the major factors considered when assessing the prognosis of breast cancer patients is whether the tumor has metastasized to distant organs. Although the exact phenotype of the malignant cells responsible for metastasis and dormancy is still unknown, growing evidence has revealed that they may have stem cell-like properties that may account for resistance to chemotherapy and radiation. One process that has been attributed to primary tumor metastasis is the epithelial-to-mesenchymal transition. In this review, we specifically discuss breast cancer dissemination to the bone marrow and factors that ultimately serve to shelter and promote tumor growth, including the complex relationship between mesenchymal stem cells (MSCs) and various aspects of the immune system, carcinoma-associated fibroblasts, and the diverse components of the tumor microenvironment. A better understanding of the journey from the primary tumor site to the bone marrow and subsequently the oncoprotective role of MSCs and other factors within that microenvironment can potentially lead to development of novel therapeutic targets.
Conflict of interest statement
Figures



References
-
- Baynes RD, Dansey RD, Klein JL, et al. High-dose chemotherapy and hematopoietic stem cell transplantation for breast cancer: past or future? Semin Oncol. 2001;28:377–388. - PubMed
-
- de Ronde J, Wessels L, Wesseling J. Molecular subtyping of breast cancer: ready to use? Lancet Oncol. 2010;11:306–307. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
